Translate this page into:
Usefulness of Pulsed Arterial Spin Labeling Magnetic Resonance Imaging in New-onset Seizure Patients and Its Comparison with Dynamic Susceptibility Contrast Magnetic Resonance Imaging
Address for correspondence: Dr. Suresh Kumar, Department of Radio-Diagnosis, IGMC, Shimla - 171 001, Himachal Pradesh, India. E-mail: thakursuresh67@yahoo.co.in
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Introduction:
Dynamic susceptibility contrast (DSC) perfusion and pulsed arterial spin labeling (PASL) imaging are newer advanced magnetic resonance sequences which are capable of detecting vascular changes in patients with new-onset seizure disorder even when no significant abnormalities are visualized on conventional sequences. The purpose of our study is to establish utility of arterial spin labeling (ASL) in new-onset seizure patients and compare ASL with DSC perfusion sequence.
Materials and Methods:
Twenty-six patients coming to emergency department with new-onset seizure disorder were evaluated using DSC and ASL sequence. Perfusion asymmetry was assessed using region of interests taken at places where signal asymmetry was maximal.
Results:
PASL sequence showed focal vascular changes in form of hyperperfusion in four patients, hypoperfusion in nine patients, and normal perfusion in 13 patients. Altered perfusion whether hypo/hyperperfusion was detected in five out of 16 patients even when conventional sequences were normal. There was strong positive linear correlation between ASL and DSC with P = 0.001.
Conclusion:
Noninvasive PASL is capable of detecting vascular changes induced by seizure and is comparable to DSC sequence. Thus, it is recommended when there is a need for repeated evaluations; in follow-up/therapy response assessment and when contrast administration is contraindicated.
Keywords
Arterial spin labeling
dynamic susceptibility contrast perfusion imaging
magnetic resonance imaging
new-onset seizure
INTRODUCTION
The first seizure is a common and very frightening event, which raises urgent health and lifestyle issues.[1] Magnetic resonance imaging (MRI) is the preferred imaging modality to detect structural abnormalities in patients with seizure because of its superior sensitivity for lesion detection.[2]
Major clinical challenge pertains to patients classified as having nonlesional epilepsy. The specificity of abnormalities seen in epilepsy has been questioned limiting its routine use in epilepsy patients.[3] Even the nonlesional seizures are associated with perfusion changes in brain which are not visualized on conventional MRI sequences.
MRI Perfusion-weighted imaging based on dynamic susceptibility contrast (DSC) permits detection of both ictal and post-ictal seizure foci by highlighting the hemodynamic changes occurring in seizure which gives results parallel to those obtained with positron emission tomography and single-photon emission computed tomography imaging. Major limitation associated with the former is that it involves intravenous contrast administration.[4] Limitations associated with the latter are the requirement of radioactive contrast material, high cost, and limited availability which have discouraged their widespread adoption in the evaluation of new-onset seizure patients.
Pulsed arterial spin labeling (PASL) MRI sequence is the latest method for perfusion-weighted imaging with the help of which cerebral perfusion and associated hemodynamic parameters can be assessed.[567] Major difference from the other older methods is the nonrequirement of exogenous contrast requirement. PASL imaging in seizure patients can be a great tool that has the potential to detect ictal and postictal perfusion changes in the form of hemispheric asymmetries noninvasively.[89]
Advanced MRI studies serve as a new diagnostic method in patients with seizure disorder. Advanced sequences, namely, perfusion-weighted images and arterial spin labeling sequences, enable detection of subtle abnormalities that may be missed by conventional MRI and thus enabling the management of seizure patients in a better way.[10]
Use of PASL sequence has been established in diseases such as cerebrovascular events and brain tumors.[1112] However, not much work has been done in the field of epilepsy. In the present study, the main objective is to analyze the usefulness in detecting vascular changes in new-onset seizure patients and to compare it with DSC-enhanced perfusion sequence.
MATERIALS AND METHODS
Study design and patient selection
We studied 26 patients (n = 26; male = 18; female = 8; ranging from 7 to 65 years with mean age = 29.73 ± 16.17 years) attending the Emergency Department in Indira Gandhi Medical College, Shimla. All patients were clinically diagnosed as case of unprovoked new-onset seizure disorder. These were classified, depending on history, into generalized tonic–clonic seizures (twenty patients), focal seizures (two patients), complex partial seizure (two patients), and focal seizure with secondary generalization (two patients).
The Institutional Review Board of Indira Gandhi Medical College, Shimla, approved the study protocol. Written informed consent from all the subjects was obtained before participation.
First episode of seizure (irrespective of type) within 48 h after onset was included in the study. Patients with cochlear implant, cardiac pacemaker, aneurysmal clips, dental filling, and any previous episode of seizure were excluded from the study.
Neurological exam
Patients were identified on clinical basis. To rule out any structural brain disease, the neurological examination was performed. In addition, the examination also extended the scope to screen for additional diseases such as pulmonary tuberculosis and hypertension and to reassure the patient, their family, and the referring doctor.
Magnetic resonance imaging acquisition
Magnetic resonance (MR) images were acquired on a 1.5T Avanto system (Siemens, Erlangen, Germany) using 18-channel head coil.
Following MRI sequences were obtained: T1 MPRAGE (TR - 1900 ms, TE = 3.25 ms, matrix size - 208 × 256, bandwidth - 203 Hz, flip angle = 8, axial section thickness - 1 mm, time of acquisition - 3:40 min), T2-weighted turbo spin-echo (TR - 3200 ms, TE - 107 ms, FOV - 230 mm2, matrix size - 218 × 320, axial section thickness - 3 mm, bandwidth - 203 Hz, number of averages - 3, flip angle - 150°, acquisition time - 2:21 min), fluid-attenuated inversion recovery (FLAIR) axial sequence (TE - 93 ms, TR - 9000 ms, FOV - 230 mm2, bandwidth - 179 Hz, time of acquisition - 2.06 min, axial section thickness - 5 mm), diffusion-weighted imaging and apparent diffusion coefficient (TR - 3400 ms, TE - 100 ms, matrix size - 154 × 192, axial section thickness - 5 mm, bandwidth - 1042 Hz, FOV - 230 mm2, acquisition time - 0.07 ms, b value - 1000), susceptibility-weighted imaging: (TR - 49 ms, TE - 40 ms, axial section thickness - 3 mm, flip angle - 15°, FOV - 230 mm2, bandwidth - 80 Hz, time of acquisition - 2:43 min), arterial spin labeling: By utilizing repeated radiofrequency pulses and proper definition of bolus length, PASL based on FAIR technique was applied. Echo-planar imaging sequence was used to acquire paired images with and without labeling utilizing: TE - 38.34 ms, TR - 4000 ms, FOV - 192 mm, matrix - 63 × 64, flip angle –180°. To minimize transit-related effects, a delay time of 1990 ms was kept between image acquisition and end of labeling pulse. DSC perfusion: It was acquired using gradient-echo echo-planar imaging utilizing TE - 47 ms, TR - 2240 ms, FOV - 230 mm, matrix - 128 × 128, flip angle –90°, slice thickness - 5 mm, and an acceleration factor (Ipat) factor of 2.5. A bolus injection of gadolinium-based contrast at a dose of 0.1 mmol/kg body weight with injection rate of 4.5 ml/s was given with a saline flush following it.
Electroencephalogram
The electroencephalogram (EEG) was obtained with a wireless 64-channel Nicolet EEG machine providing 23 electrodes positioned according to 10–20 system. 5 kΩ was kept as the limit of impedance. The EEG recording was performed for each subject in resting conditions for at least 20 min. The delay between onset of seizure and EEG ranged from 3 to 38 h (with mean = 22.92 ± 8.40 h).
Blood investigations
Blood glucose and serum electrolytes (Na, Ca) were analyzed from patients sample to rule out any metabolic cause of seizure.
Image and data analysis
Images were transferred to a separate work station where a specialized software was used to analyze the images. These were corrected for any artifacts. Conventional sequences were evaluated for any structural abnormalities.
Dynamic susceptibility contrast perfusion imaging
With the use of a postprocessing workstation (Leonardo, Syngo, Siemens, Germany), parametric maps were calculated. Relative cerebral blood flow (CBF) and cerebral blood volume (CBV) were calculated pixel by pixel, by deconvolving the measured tissue concentration time curve with an arterial input function retrieved from the middle cerebral artery branches.
Arterial spin labeling
Two circular region of interest (ROI) of about 2 cm2 were allocated symmetrically on both sides of an axial slice. ROIs were chosen such that the signal asymmetry between the two ROI was maximum. Using the mean ROI values, asymmetry index (AI) was calculated for each patient according to the formula: AIASL (%) = 100 × (ipsilateral − contralateral)/([ipsilateral + contralateral]/2). Negative and positive AI values denoted relative hypo- and hyper-perfusion in the epileptogenic focus, respectively.
The ROIs with signal asymmetry that was identified on PASL imaging were also replicated upon DSC perfusion images. For a direct comparison of PASL with DSC imaging, calculation of asymmetry indices (AICBF and AICBV) was also done on the latter.
Statistical analysis
Obtained data were analyzed using standard statistical tests. Correlation between PASL and DSC perfusion sequence was determined by calculating the Pearson's correlation coefficients between AIASL and AICBF and between AIASL and AICBV. P < 0.05 was considered statistically significant.
RESULTS
Conventional sequences revealed structural abnormalities in 10 patients (38.5%) in the form of neurocysticercosis lesions in three patients (11.5%), focal encephalomalacic changes (post trauma and stroke) in two patients (7.7%), glioma in two patients (7.7%), cerebral venous thrombosis in one patient (3.8%), tubercular lesion in one patient (3.8%), and unilateral cerebral atrophy in one patient (3.8%).
Insignificant abnormality in the form of ill-defined T2 and FLAIR hyperintensities in bilateral periventricular white matter and centrum semiovale were detected in two patients (7.7%). In the rest 14 patients (53.8%), no abnormality was detected on conventional sequences.
Based on value of asymmetrical indices calculated on DSC perfusion images, normal CBV was detected in 12 patients (46.2%), decreased CBV was noticed in nine patients (34.6%), and increased CBV was seen in five patients (19.2%). Normal CBF was seen in 14 patients (53.8%), decreased CBF was seen in eight patients (30.8%), and increased CBF was seen in four patients (15.4%).
ASL perfusion sequence was done in all the patients. Based on value of asymmetrical indices, normal CBF was seen in 13 patients (50%), decreased CBF was seen in nine patients (34.6%), and increased CBV was seen in four patients (15.4%).
On comparing CBF parameters obtained from DSC and ASL, all except one patient exhibited similar perfusion parameters in the form of normal or raised or reduced blood flow. In one patient, normal blood flow was seen on DSC; however, blood flow was reduced on ASL.
Altered CBF on both dynamic perfusion MRI and ASL was detected in five out of 16 patients despite no abnormality seen on the conventional sequences. Three of these displayed reduced CBF [Figure 1] while two patients showed increased CBF [Figure 2]. In the former three patients, MRI scan was done in early postictal period (<8 h period from the onset of seizure); on the other hand in the latter, two patients’ scan was done more than 8 h after seizure onset.
![Magnetic resonance imaging of 11-year-old male child obtained 13 h after seizure onset showing no abnormality on axial T2 (a), fluid inversion recovery (b), postcontrast – T1 (c) while revealing hypoperfusion in left frontoparietal lobe on dynamic susceptibility contrast – cerebral blood volume ([d] black asterisk), dynamic susceptibility contrast – cerebral blood flow ([e] white asterisk) and arterial spin labeling ([f] red asterisk)](/content/150/2017/8/4/img/JNRP-8-569-g001.png)
- Magnetic resonance imaging of 11-year-old male child obtained 13 h after seizure onset showing no abnormality on axial T2 (a), fluid inversion recovery (b), postcontrast – T1 (c) while revealing hypoperfusion in left frontoparietal lobe on dynamic susceptibility contrast – cerebral blood volume ([d] black asterisk), dynamic susceptibility contrast – cerebral blood flow ([e] white asterisk) and arterial spin labeling ([f] red asterisk)
![Magnetic resonance imaging of 57-year-old male child obtained 5 h after seizure onset showing insignificant T2 and fluid inversion recovery hyperintensity on axial T2 ([a] white arrow), fluid inversion recovery ([b] yellow arrow), post contrast – T1 (c) while revealing hyperperfusion in left frontoparietal lobe on dynamic susceptibility contrast – cerebral blood volume ([d] yellow asterisk), dynamic susceptibility contrast – cerebral blood flow ([e] red asterisk) and arterial spin labeling ([f] black asterisk)](/content/150/2017/8/4/img/JNRP-8-569-g002.png)
- Magnetic resonance imaging of 57-year-old male child obtained 5 h after seizure onset showing insignificant T2 and fluid inversion recovery hyperintensity on axial T2 ([a] white arrow), fluid inversion recovery ([b] yellow arrow), post contrast – T1 (c) while revealing hyperperfusion in left frontoparietal lobe on dynamic susceptibility contrast – cerebral blood volume ([d] yellow asterisk), dynamic susceptibility contrast – cerebral blood flow ([e] red asterisk) and arterial spin labeling ([f] black asterisk)
On comparing asymmetrical indices of dynamic perfusion and ASL sequences, there was linear positive correlation between the two using Pearson coefficient.
There was intermediate linear positive correlation between DSC-CBV and ASL with r = 0.560 and P = 0.003 [Figure 3].
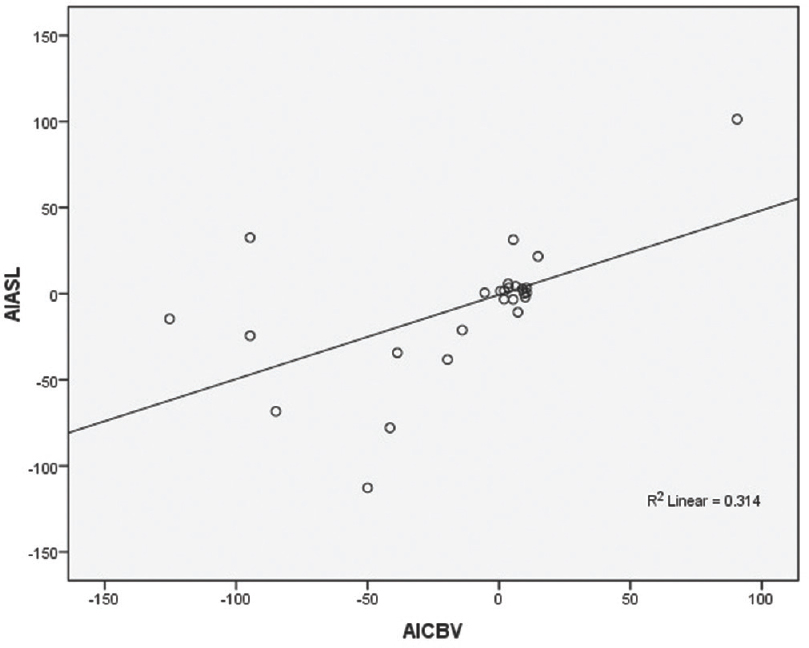
- Diagram showing Pearson correlation between the dynamic susceptibility contrast–cerebral blood volume with arterial spin labeling using their asymmetrical index
However, strong linear positive correlation was noted between DSC-CBF and ASL with r = 0.766 and P = 0.001 [Figure 4].
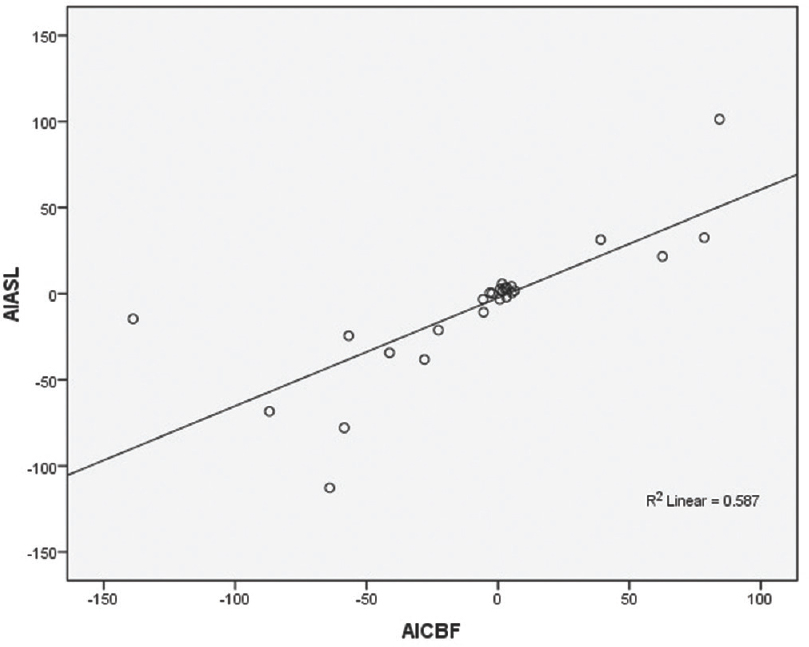
- Diagram showing Pearson correlation between the dynamic susceptibility contrast–cerebral blood flow with arterial spin labeling using their asymmetrical index
DISCUSSION
Epilepsy is among the most common serious neurologic disorder. In India, the prevalence rate of epilepsy is around 5.59/1000 population.[13] Globally, the prevalence in developed countries is between 5 and 10/1000 population.[14] In developed countries, the incidence is around 50/100,000/year.[14]
Seizure episode leads to increase in metabolic demand in an involved cerebral cortex. It is associated with temporary increase in regional brain blood flow. Therefore, for localization of seizure focus by imaging, evaluation of cerebral perfusion has become a widely accepted method.[15]
Earlier nuclear medicine imaging techniques were used to detect these perfusion changes. Now, MRI is routinely used to figure out potential structural disorders in seizure patients.[16] Newer MR sequences such as DSC have made possible detection of these cerebral perfusion changes.[17] Arterial spin labeling, which is a developing MR perfusion technique, has the ability of quantifying local relative CBF by measuring the inflow of electromagnetically labeled arterial blood into the target area and does not require contrast medium administration.[18] In this technique, water molecules in the neck are inverted by applying radiofrequency pulses. These inverted or saturated water molecules are imaged after a certain delay time which allows the labeled water molecules to reach the capillary vasculature in the brain and exchange with water molecules in the brain tissue. ASL MRI thus consists of two phases: the preparation phase, in which inflowing blood is labeled, and the acquisition phase, in which perfusion-weighted images are acquired. By alternating between the acquisition of labeled images and control images, which only differ in the magnetized state of arterial blood, paired label-control images are obtained. A subtraction image, control image minus label image, reveals the perfusion signal.
ASL also known as spin tag perfusion imaging is a MRI method that measures quantitative CBF. The advantages of ASL compared with conventional perfusion techniques include repeatability, absolute quantification, and the avoidance of intravenous contrast administration.
We performed ASL sequence to evaluate vascular and perfusion abnormalities in new-onset seizure patients. In our study, we found that perfusion abnormalities were seen in the form of either hypo- or hyper-perfusion in patients with a structural cause of epilepsy and similar changes were also seen in patients in whom no abnormality was detected on conventional sequences. These findings have been demonstrated by previous studies done by Pollock et al., Pendse et al., Matsuura et al., and Kim et al.[15192021] In addition, our study advocates the fact that hyperperfusion is observed when MRI scan is done in early postictal period (i.e., <8 h) compared to hypoperfusion changes seen in late postictal period (more than 8 h). These changes were consistent with previous studies done by Pizzini et al. and Storti et al.[1022] In some patients, no vascular changes were seen in spite of a structural lesion though their proportion was very less.
Strong positive linear correlation between DSC-CBF and ASL along with intermediate positive linear correlation between DSC-CBV and ASL observed in our study substantiate the fact that ASL is equally valuable as DSC perfusion imaging in detecting perfusion abnormalities in seizure patients. These findings are in concordance with earlier studies done by Pizzini et al. and Oner et al.[1023] Thus, DSC perfusion can be replaced by ASL sequence, especially in conditions with contraindications to contrast administration such as renal insufficiency.
Previous original research on ASL sequence in seizure patients has included diagnosed patients of epilepsy who were on medical treatment. Our study included patients coming with first episode of seizure with no history of antiepileptic medication in past. Our study had few limitations, that is, we studied a small sample size as incidence of new seizure patients is relatively less and most of these patients could not avail medical facilities early owing to difficult topographic location of our medical institute.
CONCLUSION
Seizure episodes are associated with perfusion alterations within brain parenchyma. Conventional MRI sequences are able to detect only structural abnormalities which lead to seizure. However, advanced MRI sequences such as DSC-enhanced MRI and ASL sequences can detect these perfusion changes which are not detected on conventional sequences. ASL is as good as dynamic perfusion sequence in detecting increased or decreased cerebral perfusion with a major advantage that no exogenous contrast injection is needed in comparison to dynamic sequence in which exogenous contrast injection is required. However, further research with larger sample size is required to validate the above finding. Asymmetrical indices of ASL show positive linear correlation with those of dynamic perfusion sequence. Thus, ASL can obviate the need of exogenous contrast to study cerebral perfusion in new-onset seizure patients and can be used in conditions where contrast injection is contraindicated.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- First seizure: EEG and neuroimaging following an epileptic seizure. Epilepsia. 2008;49(Suppl 1):19-25.
- [Google Scholar]
- Neuroimaging of first-ever seizure Contribution of MRI if CT is normal. Neurol Clin Pract. 2013;3:398-403.
- [Google Scholar]
- Clinical applications of MR spectroscopy in epilepsy. Neuroimaging Clin N Am. 2004;14:507-16.
- [Google Scholar]
- Comparative overview of brain perfusion imaging techniques. Stroke. 2005;36:e83-99.
- [Google Scholar]
- Arterial spin-labeling in routine clinical practice, part 1: Technique and artifacts. AJNR Am J Neuroradiol. 2008;29:1228-34.
- [Google Scholar]
- Arterial spin-labeling in routine clinical practice, part 2: Hypoperfusion patterns. AJNR Am J Neuroradiol. 2008;29:1235-41.
- [Google Scholar]
- Arterial spin-labeling in routine clinical practice, part 3: Hyperperfusion patterns. AJNR Am J Neuroradiol. 2008;29:1428-35.
- [Google Scholar]
- Usefulness of pulsed arterial spin labeling MR imaging in mesial temporal lobe epilepsy. Epilepsy Res. 2008;82:183-9.
- [Google Scholar]
- Cerebral perfusion alterations in epileptic patients during peri-ictal and post-ictal phase: PASL vs. DSC-MRI. Magn Reson Imaging. 2013;31:1001-5.
- [Google Scholar]
- Magnetic resonance perfusion imaging in acute ischemic stroke using continuous arterial spin labeling. Stroke. 2000;31:680-7.
- [Google Scholar]
- A comparative study of perfusion measurement in brain tumours at 3 Tesla MR: Arterial spin labeling versus dynamic susceptibility contrast-enhanced MRI. Eur Neurol. 2010;64:21-6.
- [Google Scholar]
- Capability of arterial spin labeling MR imaging in localizing seizure focus in clinical seizure activity. Eur J Radiol. 2016;85:1295-303.
- [Google Scholar]
- Diffusion and perfusion MRI for the localisation of epileptogenic foci in drug-resistant epilepsy. Neuroradiology. 2002;44:475-80.
- [Google Scholar]
- Arterial spin-labeled magnetic resonance imaging in hyperperfused seizure focus: A case report. J Comput Assist Tomogr. 2008;32:291-2.
- [Google Scholar]
- Interictal arterial spin-labeling MRI perfusion in intractable epilepsy. J Neuroradiol. 2010;37:60-3.
- [Google Scholar]
- Usefulness of arterial spin-labeling images in periictal state diagnosis of epilepsy. J Neurol Sci. 2015;359:424-9.
- [Google Scholar]
- Combining ESI, ASL and PET for quantitative assessment of drug-resistant focal epilepsy. Neuroimage. 2014;102(Pt 1):49-59.
- [Google Scholar]
- pASL versus DSC perfusion MRI in lateralizing temporal lobe epilepsy. Acta Radiol. 2015;56:477-481.
- [Google Scholar]






