Translate this page into:
Spinal shortening and monosegmental posterior spondylodesis in the management of dorsal and lumbar unstable injuries
Address for correspondence: Dr.Tarek A Aly, 48th, Sarwat Street, Tanta - 31111, Egypt. E-mail: tahmed21@hotmail.com
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Introduction:
Patients with spinal injuries have been treated in the past by laminectomy in an attempt to decompress the spinal cord. The results have shown insignificant improvement or even a worsening of neurologic function and decreased stability without effectively removing the anterior bone and disc fragments compressing the spinal cord. The primary indication for anterior decompression and grafting is narrowing of the spinal canal with neurologic deficits that cannot be resolved by any other approach. One must think of subsequent surgical intervention for increased stability and compressive posterior fusion with short-armed internal fixators.
Aim:
To analyze the results and efficacy of spinal shortening combined with interbody fusion technique for the management of dorsal and lumbar unstable injuries.
Materials and Methods:
Twenty-three patients with traumatic fractures and or fracture-dislocation of dorsolumbar spine with neurologic deficit are presented. All had radiologic evidence of spinal cord or cauda equina compression, with either paraplegia or paraparesis. Patients underwent recapping laminoplasty in the thoracic or lumbar spine for decompression of spinal cord. The T-saw was used for division of the posterior elements. After decompression of the cord and removal of the extruded bone fragments and disc material, the excised laminae were replaced exactly in situ to their original anatomic position. Then application of a compression force via monosegmental transpedicular fixation was done, allowing vertebral end-plate compression and interbody fusion.
Results:
Lateral Cobb angle (T10–L2) was reduced from 26 to 4 degrees after surgery. The shortened vertebral body united and no or minimal loss of correction was seen. The preoperative vertebral kyphosis averaged +17 degrees and was corrected to +7 degrees at follow-up with the sagittal index improving from 0.59 to 0.86. The segmental local kyphosis was reduced from +15 degrees to −3 degrees. Radiography demonstrated anatomically correct reconstruction in all patients, as well as solid fusion.
Conclusion:
This technique permits circumferential decompression of the spinal cord through a posterior approach and posterior interbody fusion.
Keywords
Injury
laminoplasty
spine
shortening
spondylodesis
Introduction
With an average incidence of 11,000 cases per year in the United States, spinal cord injury (SCI) is a significant cause of morbidity and mortality,particularly among teenagers and young adults. The main causes of acute SCI are traffic accidents, sports and recreational activities, accidents at work, falls in the home, and violence.[1] Because there is a lack of effective treatment for restoring neurologic function below the level of SCI, the vast majority of SCI victims face many years of losing independence and continued medical expenses.
It is now well recognized that laminectomy as the sole surgical technique is contraindicated in most cases of acute SCI because it usually fails to produce adequate decompression of the spinal cord and often causes spinal instability that lead to neurologic deterioration. Recent advances in the safety and efficacy of spinal cord surgical decompressive procedures offer significant potential for repairing some of the neurological damage caused by injuries to the spine.[2–6]
The aim of this study was to analyze the results and efficacy of the recapping laminoplasty in which the posterior elements are removed and replaced combined with interbody fusion technique for decompression and fusion of dorsal and lumbar unstable injuries.
Materials and Methods
Twenty-three patients with fractures and/or fracture-dislocations of dorsal and lumbar spine with neurologic deficit are presented. They were 16 males and 7 females. Fall from height was the cause of injury in most of the cases. The most frequently injured segment was T11–L1. All patients underwent surgery within 1 week after the injury except one who had surgery after 2 months.
All patients had radiologic evidence of spinal cord or cauda equina compression, with either paraplegia or paraparesis. Neurologic assessment was performed with use of a grading scale of Frankel et al., as modified by Bradford and McBride[19] as follows: (A) Complete motor and sensory loss. (B) Preserved sensation only, voluntary motor function absent. (C) Preserved motor less than fair grade (nonfunctional). (D1) Preserved motor at lowest functional grade (3+/5+) and/or with bowel or bladder paralysis with normal or reduced voluntary motor function. (D2) Preserved motor at mid-functional grade (3+ to 4+/5+) and/or neurologic bowel or bladder dysfunction. (D3) Preserved motor at high functional grade (4+/5+) and normal voluntary bowel or bladder function. (E) Complete normal motor and sensory function (may still have abnormal reflexes). There were 4 patients graded A, 3 patients graded B, 2 graded D1, and 14 patients graded D3.
The patients were classified according to the three-columns theory of Denis.[7] The kyphotic and scoliotic angles were measured according to the Cobb method.[8] The narrowing of the spinal canal at the fracture level (N%) was estimated by measuring the diameter in the mid-sagittal plane (F) and comparing it with the mean value of mid-sagittal diameters at the vertebra above (A) and below (B), according to the formula[9]: N = 100 – F/A+B/2 × 100.
Operative technique
Standard posterior mid-line approach was used. The paraspinal muscles are dissected, leaving the supra- and interspinous ligaments intact. Subperiosteal exposure of the posterior elements is performed to bases of the transverse processes. Undue injury to the facet capsules should be avoided.
To prepare the pathway for the T-saw (flexible multifilament thread wire saw, 0.54 mm in diameter)[10] guide under the lamina and through the nerve root canal, the soft tissue attached to the lateral aspect of the pars interarticularis is dissected and removed using utmost care so as not to damage the exiting nerve root. The inferior half of the spinous process of the superior adjacent vertebra of the fractured one is excised by the T-saw, while leaving the interspinous ligament intact. The ligamentum flavum is excised bilaterally. The C-curved malleable T-saw guide then is introduced through the interlaminar space along the course of the exiting nerve root in a cephalocaudal direction. The tip of the T-saw guide can be found ventral to the inferior extent of the pars interarticularis at the exit zone of the neural foramen.
Next, a T-saw is passed through the hole in the T-saw guide. The T-saw guide is withdrawn as tension is maintained on the T-saw with a T-saw holder at each end [Figure 1]. Using a reciprocating motion, the saw produces a fine cut of the pars interarticularis. Then the facet capsules are divided sharply, and the caudal ligamentum flavum is excised. The lamina then may be rotated out of the surgical field [Figure 2]. Care must be taken to lubricate the T-saw continuously with saline to avoid excessive heat from friction and to wash out bone debris.
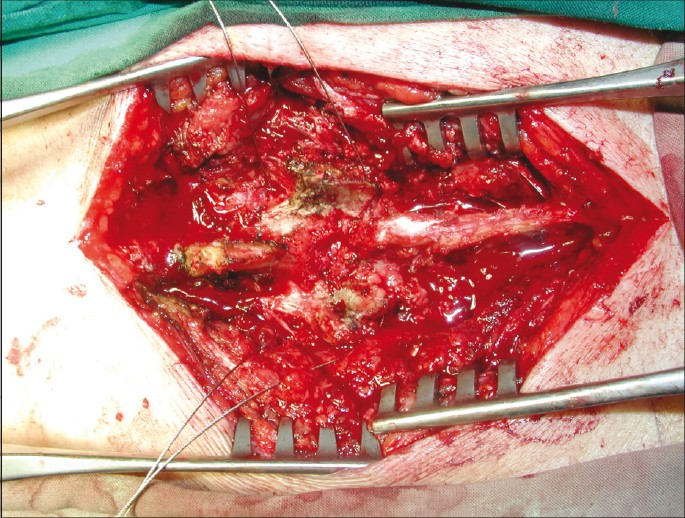
- A photo showed the T-saw passed through the nerve root canal on both sides
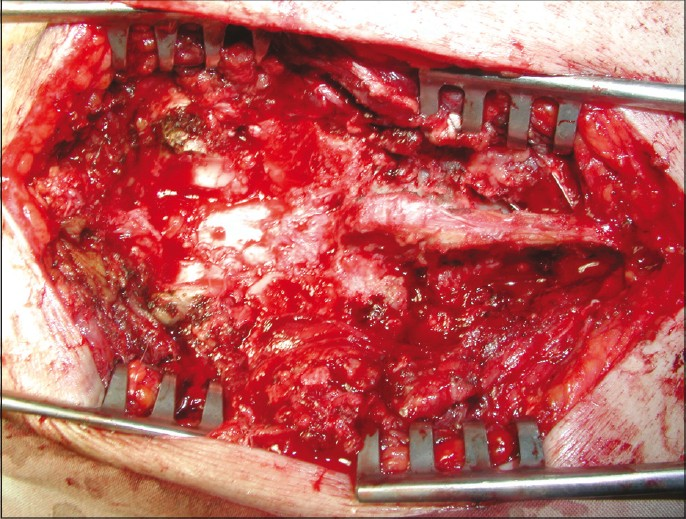
- A photo showed the surgical field after the lamina was removed in one piece. The operative field was so wide that it enabled us to manipulate the spine anteriorly and removing the disc material with the retropulsed fragment
Decompression of the cord and compromised nerve root was performed by removal of the extruded bone fragments and disc material in case of burst fractures, reduction in case of dislocations. Then, the posterior arches are reconstructed. Because the bone loss with the T-saw cut is minimal and smooth, the excised lamina can be restored to their exact original anatomic position. Then application of a compression force via monosegmental transpedicular fixation was done, allowing vertebral end plate compression and interbody fusion [Figure3 and 4].
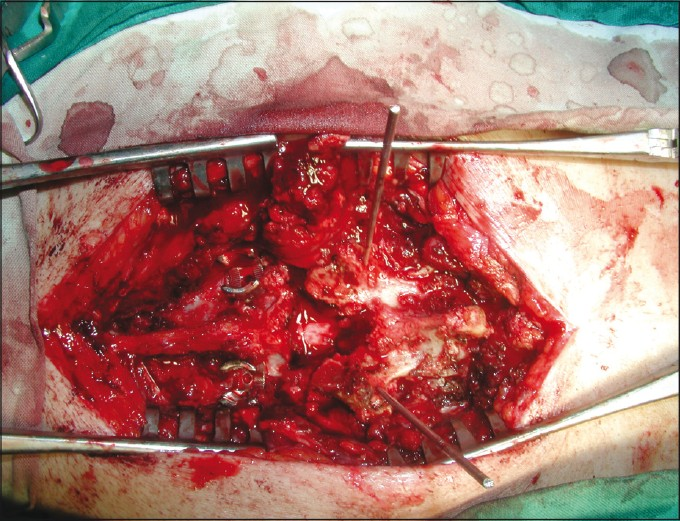
- A photo showed the lamina in its place fixed temporarily with two K wires
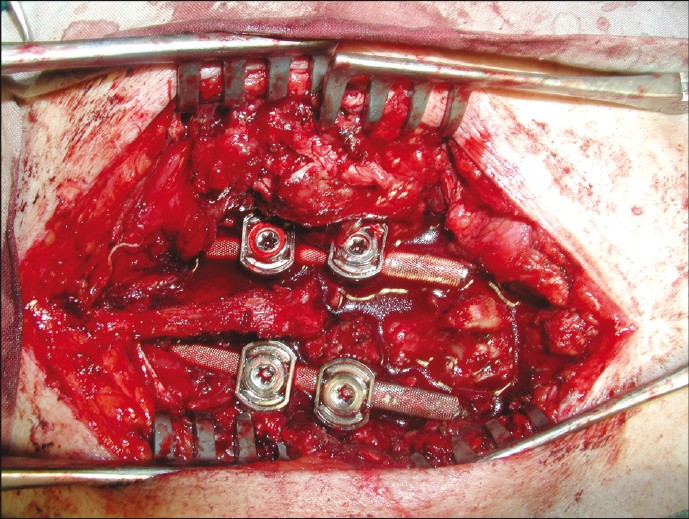
- A photo showed the final transpedicular fixation
Postoperative care
All patients were fitted with a polypropylene thoracolumbosacral orthosis. Orthosis were worn for approximately 3 months and the decision to wean was made on an individual basis.
Follow-up study
The average follow-up period was 67 months (range, 38–96 months). The clinical outcome was assessed by analysis of Frankel grading.
Results
The mean age was 42 years (range, 18–53 years). There were 16 males and 7 females. Eighteen injuries were located in the thoracolumbar region (T11–L1) and 5 injuries were located in the lumbar region (L2–L3). Associating injuries were frequent; head injury had occurred in 6 patients, abdominal injury in 1, and calcaneal fracture in 7 patients. The mean time from the initial incision to dressing application was 108 min (range, 100–124 min). The mean volume of the blood loss was 610 cc (range, 410–860 cc). There were 4 patients graded A, 3 patients graded B, 2 graded D1, and 14 patients graded D3. The patients with incomplete neurologic deficits improved by at least one grade, while patients with grades A and B showed no recovery.
During surgery, sufficient visual field was obtained. The T-saw did not cause any dural tears. There were no wound infections or other complications noted. Cutting of the posterior bony elements using T-saw cause 2–4 mm loss of height, which allow shortening using posterior transpedicular compression device.
After surgery, the spinal canal of all patients had increased, with mean initial encroachment of 40% (range, 30%–70%) decreasing to 12.8% after surgery (range, 7%–32%). The gain in the canal size was larger at T12 and L1 than L2 level. Compared with preoperative values, postoperative encroachment was reduced by 66% that mean better canal restoration.
The initial kyphosis, scoliosis, and initial compression associated with fractures at the thoracolumbar and lumbar levels were assessed to determine the different degrees of correction that can be achieved. Lateral Cobb angle (T10–L2) was reduced from 26 to 4 degrees after surgery. The preoperative vertebral kyphosis averaged +17 degrees and was corrected to +7 degrees at follow-up with the sagittal index improving from 0.59 to 0.86. The segmental local kyphosis was reduced from +15 degrees to −3 degrees. The shortened vertebral body united and no loss of correction was seen [Figure5 and 6].
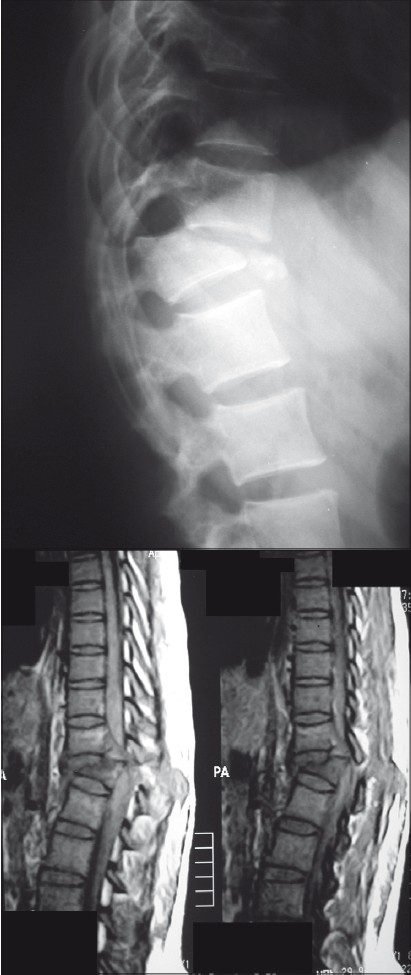
- A plain and MRI radiograph of a female patient aged 29 years with fracture-dislocation at T11– T12 level with complete paraplegia
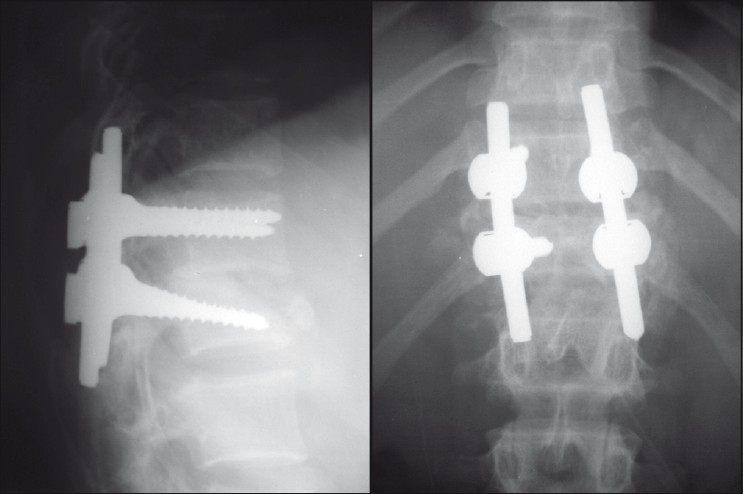
- Postoperative radiograph of the same patient showed monosegmental fixation with transpedicular screw fixation
Discussion
In unstable thoracolumbar fractures, the spinal deformity consists of a loss of vertebral body height, kyphotic angulation, and retropulsion of bone into the spinal canal. The value of decompression of the spinal canal in acute spinal injuries, whether anterior or posterior, has not been clearly demonstrated, although there is an empirical impression that clearance of the spinal canal may improve neurologic recovery in some patients with an incomplete neurologic deficit.[11]
Posterior instrumentation restores vertebral body height via distraction forces. Additionally, the anterior and middle columns are restored to normal length during correction of kyphosis. The spinal column assumes the contour of the rods, resulting in correction of kyphotic deformity. Indirect spinal canal decompression is thought to be achieved by development of tension in the posterior longitudinal ligament, which then pushes the retropulsed bone fragments forward. This process has been termed ligamentotaxis,[12] Fredreickson et al.[13] demonstrated that the posterior longitudinal ligament had little effect in vitro, but rather the tension created by the annulus fibrosus of the disc as a result of the distraction reduced retropulsed bone fragments. Clinical studies documenting incomplete reductions of the spinal canal by posterior instrumentation. The posterior distraction instrumentation reduces canal compromise by only 50% of the initial occlusion, leaving an average of 32% canal compromise.[1415] Therefore, neurologic recovery is uncertain as consistent neurologic recovery was noted if a canal patency of more than 66% of the patient's normal canal space was achieved.[16]
Flesh et al.[17] described posterolateral decompression technique that allows forward impaction of the retropulsed bone and disc fragments, thus decompressing the ventral side of the neural tissues. This technique is facilitated by the removal of the facets or pedicles. Manipulation and medial retraction of the neural elements in the injury zone and copious epidural bleeding are potential drawbacks of this approach. There were also the bilateral transpedicular decompression techniques that gave an average improvement of 0.9 Frankel grades per patient.[18] Bony fusion is difficult to achieve in patients treated with posterolateral decompression because of the large surgically created bony defect.[19]
On the other hand, direct anterior decompression of the spinal canal provides an optimal environment for recovery of the neural tissue. In the mean time, anterior decompression and strut grafting of the acute fractures causes a significant increase of instability and the possible development of a progressive deformity and graft dislodgment.[20] Therefore, anterior instrumentation has been developed but proved too bulky and was associated with late vascular complications.[21] The residual canal stenosis with anterior decompression was significantly lower but no difference in neurologic outcome was reported compared with the posterior decompression.[2223]
Short segment posterior fixation and fusion in case of thoracolumbar fractures or fracture dislocations were proved by many authors. Razak et al.[24] stated that no loss of reduction or of fixation was noted in any of his patients and solid fusion was achieved in all patients. Gelb et al.[25] concluded that with modern instrumentation and techniques, short-segment thoracolumbar fracture fixation could be used successfully, despite highly comminuted injuries, without anterior column support or supplemental bracing.
Therefore, we advocated our technique to obviate the complication of the anterior surgery and get its advantages of good decompression of the spinal cord and nerve roots and in the mean time get the advantages of good stability and better fusion of the posterior technique. Recapping laminoplasty was firstly reported by Kawahara et al[26] who treated his patients with spinal cord tumors by a unique posterior method of removing and replacing the posterior spinal elements using T-saw (“recapping T-saw laminoplasty”). He reported good decompression of the cord by this technique.
Using recapping laminoplasty in management of thoracolumbar injuries was not used by many surgeons all over the world. The Medline search reveals that only one author[27] used this technique and he stated that this is an ideal operation method for treating burst fractures of thoracolumbar vertebra with spinal stenosis, which is simple, safe, and enlarging vertebral canal, at the same time completely decompressing the dura mater and nerve roots. This method eliminated secondary spinal stenosis, reconstructed posterior column, and increased spinal stability.
Comparing our early results to the previously reported ones,[319] it was similar to that described in series with anterior decompression, fusion, and instrumentation. The neurologic improvement of at least one Frankel grade was achieved. Also, we had the same spinal canal clearance. Compared with anterior decompression and stabilization, this procedure is technically less demanding, carries low risks, and ensures good stabilization.
Previous reports[2829] suggest that transpedicular discectomy and grafting of the upper injured disc space may be more effective based on the assumption that a substantial amount of correction loss occurs at the injured disc space. Therefore, discectomy and monosegmental fusion provide better results with good bony fusion anteriorly and no loss of correction obtained.
A number of reports also have described the biomechanical importance of the posterior components, such as lamina, facet joints, supra- or interspinous ligament, or ligamentum flavum.[30] Therefore, anatomic replacement of the posterior elements is the optimal strategy, if technically possible.
The T-saw is a device for cutting the bone, which was originally developed for safely cutting the pedicle in total en bloc spondylectomy.[31] It has a number of advantages.
The T-saw is thin and flexible and can be introduced safely into a confined space, such as the epidural space or the intervertebral foramen, with the use of the T-saw guide. The surface of the T-saw is so smooth that it is possible to cut the lamina or the pedicle without injuring the dura matter or the spinal nerve roots; moreover, the saw does not jam in the bone during use. Also, the direction of the cut is from ventral to dorsal away from the meninges and the spinal cord. None of patients had dural tears, damage to the nerve roots, or leakage of cerebrospinal fluid.
The cut of the T-saw is so thin that the bone loss is negligible, which made it possible to achieve exact matching when replacing the lamina. Primary bone healing with no excessive bone formation might occur.[32]
Conclusions
The recapping laminoplasty combined with interbody fusion technique effectively restores the 3 columns of the spine, the anterior column through interbody bony fusion, the middle column through effective removal of the retropulsed disc fragment and the transpedicular fixation with restoration of the sagittal alignment, and the posterior column through anatomic reconstruction of the posterior arches and preservation of their biomechanical function. Recapping laminoplasty combined with interbody fusion technique offers better canal clearance similar to the anterior decompression technique but technically less demanding, carries low risks, and ensures good stabilization.
Source of Support: Nil
Conflict of Interest: None declared.
References
- An evidence-based review of decompressive surgery in acute spinal cord injury: Rational, indications, and timing based on experimental and clinical studies. J Neurosurgery (Spine 1). 1999;91:1-11.
- [Google Scholar]
- Comparison of two types of surgery for thoraco- lumbar burst fractures: Combined anterior and posterior stabilization vs.posterior instrumentation only. Acta Neurochir (Wien). 1999;141:349-57.
- [Google Scholar]
- Minimally invasive retroperitoneal approach for lumbar corpectomy and anterior reconstruction.Technical note. J Neurosurg. 2000;93:161-7.
- [Google Scholar]
- Posterior spinal shortening for paraplegia after vertebral collapse caused by osteoporosis. Spine. 2000;25:2832-5.
- [Google Scholar]
- Selection criteria and outcome of operative approaches for thoracolumbar burst fractures with and without neurological deficit. J Neurosurg. 1997;86:48-55.
- [Google Scholar]
- Anterolateral decompression for traumatic spinal cord compression. J Pak Med Assoc. 1994;44:242-3.
- [Google Scholar]
- Outline for study of scoliosis.Instructional course lectures. Ann Arbor, Edwards. The American Academy Of Orthopedic Surgery. 1984;5:251-275.
- [Google Scholar]
- Burst fractures in the thoracic and lumbar spine.A clinico-neurologic analysis. Spine. 1989;14:1316-23.
- [Google Scholar]
- The thread wire saw: A new device for cutting bone. J Bone Joint Surg (Am). 1996;78:1915-7.
- [Google Scholar]
- Canal clearance in burst fractures using the AO internal fixator. Spine. 1992;17:558-60.
- [Google Scholar]
- Stabilization of the lower thoracic and Lumbar spine with the internal skeletal fixation system: Indications, techniques, and first results of treatment. Spine. 1987;12:544-551.
- [Google Scholar]
- Vertebral burst fractures: an experimental, morphologic, and radiographic study. Spine (Phila Pa 1976). 1992;17:1012-21.
- [Google Scholar]
- Spinal canal clearance in burst fractures using the AO Internal Fixator. Orthop Trans. 1991;15:318.
- [Google Scholar]
- Indirect spinal canal decompression in patients with thoracolumbar burst fractures treated by posterior distraction rods. J Spinal Disord. 1991;4:39-48.
- [Google Scholar]
- Early rod-sleeve stabilization of the injured thoracic and lumbar spine. Orthop Clin North Am. 1986;17:121-45.
- [Google Scholar]
- Harrington instrumentation and spine fusion for unstable fractures-dislocations of the thoracic and lumbar spine. J Bone Joint Surg. 1977;59:143-53.
- [Google Scholar]
- Bilateral Transpedicular decompression and Harrington rod stabilization in the management of severe thoracolumbar burst fractures. Spine. 1992;17:162-71.
- [Google Scholar]
- Surgical management of thoracolumbar spine fractures with incomplete neurologic deficits. Clin Orthop. 1987;218:201-16.
- [Google Scholar]
- Thoracolumbar spine fractures with neurologic deficit. Orthop Clin North Am. 1994;25:595-612.
- [Google Scholar]
- Does ‘canal clearance” affect neurological outcome after thoracolumbar burst fractures? J Bone Joint Surg (Br). 2000;82:629-35.
- [Google Scholar]
- Thoracolumbar fracture-dislocation results of surgical treatment. Med J Malaysia. 2000;55:14-7.
- [Google Scholar]
- Successful Treatment of Thoracolumbar Fractures With Short-segment Pedicle Instrumentation. J Spinal Disord Tech. 2010;23:293-301.
- [Google Scholar]
- Recapping laminoplasty for the treatment of severe burst fractures of thoracolumbar vertebra complicated with spinal stenosis. Zhongguo Gu Shang. 2008;21:445-6.
- [Google Scholar]
- Burst fractures with neurologic deficits of the thoraco-lumbar spine: Results of anterior decompression and stabilization with anterior instrumentation. Spine. 1984;9:788-95.
- [Google Scholar]
- Application of posterior plating and modification in thoracolumbar spine injuries. Spine. 1991;16:S125-33.
- [Google Scholar]
- Treatment of thoracolumbar burst fractures without neurological deficit by indirect reduction and posterior instrumentation: Bisegmental stabilization with monosegmental fusion. Eur Spine J. 1999;8:284-9.
- [Google Scholar]
- The significance of the vertebral posterior elements in the mechanics of the thoracic spine. Clin Orthop. 1971;81:2-14.
- [Google Scholar]
- Total enbloc spondylectomy: A new surgical technique for primary malignant vertebral tumors. Spine. 1997;22:324-33.
- [Google Scholar]






