Translate this page into:
Endoscopic intradural extramedullary lesion excision: Results of augmented duroplasty with artificial dura
*Corresponding author: Anant Mehrotra, Department of Neurosurgery, Sanjay Gandhi Postgraduate Institute of Medical Sciences, Lucknow, Uttar Pradesh, India. dranantmehrotra@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Mehrotra A, Kumar A, Raiyani V, Singh R, Verma PK, Das KK, et al. Endoscopic intradural extramedullary lesion excision: Results of augmented duroplasty with artificial dura. J Neurosci Rural Pract 2023;14:177-81.
Abstract
Objectives:
Prevailing techniques of dural closure in endoscopic spine surgery (ESS) for intradural extramedullary (IDEM) tumor excision increase the steep learning curve and operative time. We aimed to assess the efficacy of augmented duroplasty with artificial dura and share our initial experience of ESS for IDEM excision.
Materials and Methods:
We retrospectively analyzed 18 (n = 18) consecutive patients of IDEM tumors operated by ESS using Destandau’s endoscopic system. The pre-operative, post-operative, and at the latest follow-up clinical status were recorded in terms of Nurick’s grades and the Oswestry Disability Index. Immediate post-operative complications and intraoperative findings were noted from hospital information system and patient records.
Results:
The mean (± SD) age of patients was 40.3 ± 14.9 (range 19–64) years, with M: F ratio of 2:1. All the lesions were intradural and present at lumber (n = 6), thoracic (n = 9), and cervical (n = 3) regions. The average duration of surgery, blood loss, hospital stay, and duration of follow-up were 157 ± 45.3 (90–240) min, 168.8 ± 78.8 (30–300) mL, 4.29 ± 1.4 (2–7) days, and 19.3 ± 7.2 (7–36) months, respectively. There were no CSF leaks, wound-related complications, or material-induced adverse events.
Conclusion:
In endoscopic IDEM excision, dural closure with artificial dura is efficient in preventing CSF leak. It shortens the steep learning curve and improves the surgical outcome due to technical ease.
Keywords
Artificial dura
Intradural extramedullary tumor
CSF leak
Endoscopic spine surgery
Minimally invasive surgical technique
INTRODUCTION
Minimally invasive surgical (MIS) approaches attempt to preserve the normal myo-ligamento-osseous complex. MIS techniques are associated with lesser blood loss, shorter post-operative stay, reduced use of narcotics, and better biomechanical stability.[1-4] The endoscopic technique for the excision of IDEM lesions is continuously evolving. Achieving watertight dural closure is one of the major challenges in minimally invasive procedures. Multiple studies have reported different techniques of dural closure in minimally invasive resection of IDEM lesions.[5-10] However, there is no study reporting the impact of using artificial dural substitute in MISS IDEM excision using endoscope with Destandau’s retractor. In this study, we aimed to assess the feasibility of adequate dural closure with artificial dura in the endoscopic excision of IDEM and its possible role in surgical outcome.
MATERIALS AND METHODS
We retrospectively analyzed 18 (n = 18) consecutive patients of IDEM operated by endoscopic spine surgery (ESS) using Destandau’s endoscopic system by a single surgeon (AM) in the year 2019–2022. Patients were included irrespective of the pathology or the vertebral level of the lesion. Patients who were not suitable for the excision by endoscopic Destandau’s technique, those who required an operating microscope at any stage of the surgery, or those who did not consent for the procedure were excluded from the study. The clinical assessment was done using Nurick’s grade and the Oswestry Disability Index (ODI). Preoperatively, an X-ray and magnetic resonance imaging (MRI) were done. The post-operative MRI was done within 48 h of surgery or after 6 weeks. Immediate post-operative complications and intraoperative findings were noted from hospital information system and patient records. The patients were followed up in OPD and the final clinical status (Nurick’s grade and ODI scores) was reported from the latest follow-up.
Operative technique
Docking
The target level along with midline (spinous process) was marked under fluoroscopy. A 1.5 cm paraspinal (1–1.5 from midline) incision was made on the side of the lesion. The thoracolumbar fascia was incised. It was followed by subperiosteal elevation of paraspinal muscles along the spinous process and lamina, till the medial border of the facet joint laterally. The Destandau’s system was docked at the desired interlaminar space and the level was confirmed again under C-arm.
Bony exposure
Under the endoscopic view, the adjacent hemi-lamina and root of the spinous process were drilled using 3 mm cutting drill bit to expose the dura.
Dural opening and tumor excision
Dura was opened. The upper and the lower end of the tumor was identified (poles of the tumor were identified by a “rounded” feel, increase in the egress of CSF, and by direct visualization). The proximal and distal attachment of the lesion was identified, coagulated, and cut. The tumor was delivered out from the dural opening and taken out through the port. In the case of large tumors, initial piecemeal removal of the tumor was done, and subsequently, the poles were identified and the stalk or attachment identified, coagulated, and cut followed by excision of the tumor.
Dural closure
After achieving hemostasis, artificial dura, measuring about 1 × 0.5 cm, was folded and inserted through the instrument port. It was laid over the dural defect and opened. The edges were tucked under the surrounding bone at the margins. The Destandau’s system was removed. The fascia closed with interrupted absorbable sutures and the skin closed with subcuticular sutures. Figure 1 and 2 are illustrative images of a case of D6 meningioma operated with this technique.
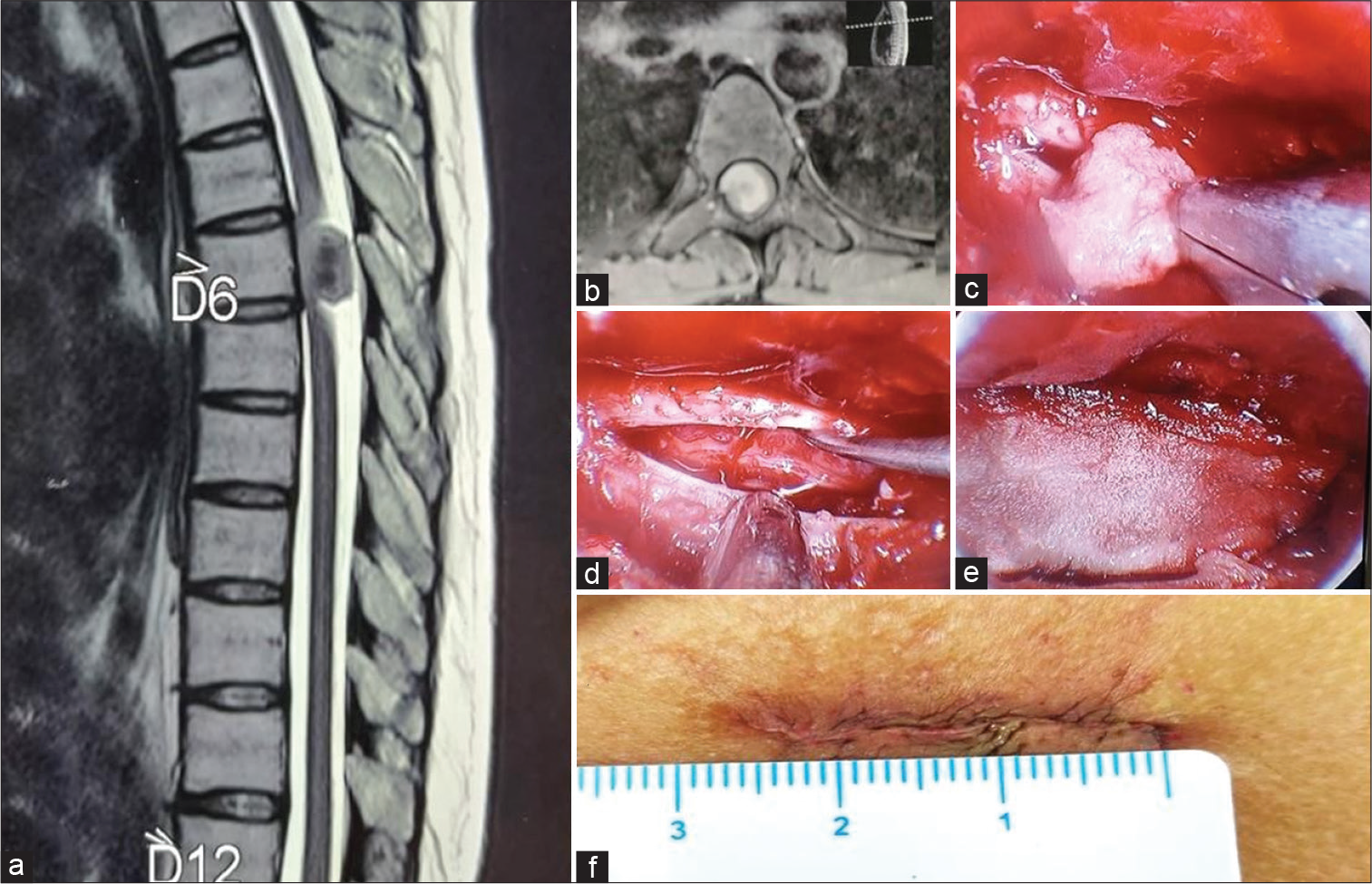
- (a) T2-weighted magnetic resonance imaging (MRI), sagittal section, showing well-defined ventrally placed intradural extramedullary lesion at D6 vertebral body level. (b) T1-weighted contrast-enhanced MRI, axial section, showing a homogeneous contrast-enhancing IDEM lesion. (c) Intraoperative image: Tumor excision. (d) Dural opening with cord seen within. No residual tumor is seen. (e) Artificial dura was placed over the dural defect. (f) Sutured incision (2.5 cm).
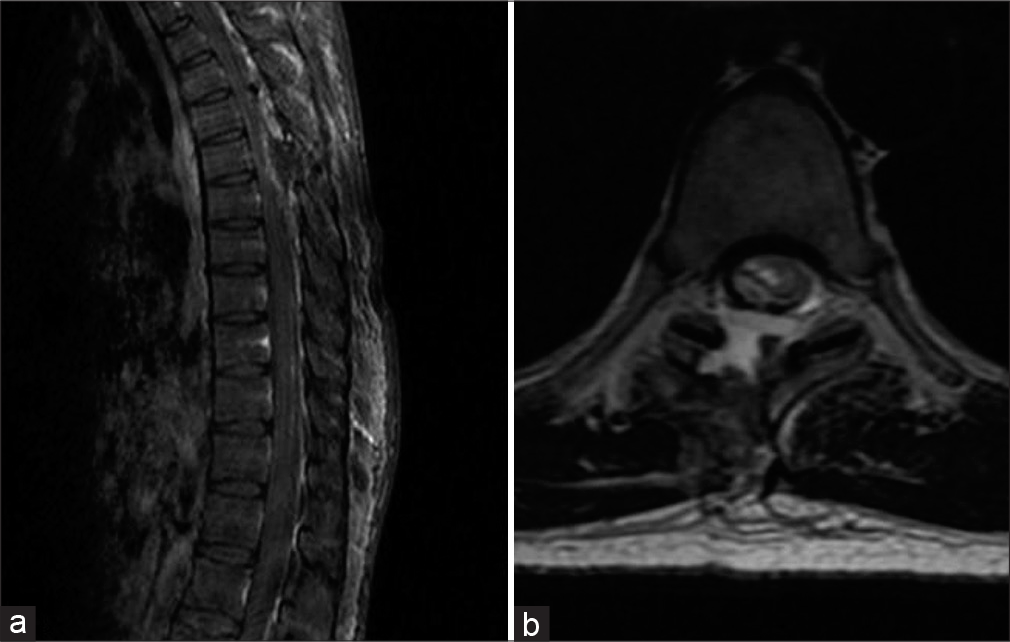
- Immediate post-operative MRI of the patient with D6 vertebral body level meningioma. (a) T1 sagittal contrast-enhanced MRI. (b) T2 axial MRI.
Statistical analysis
Statistical analysis was done using Statistical Package for the Social Sciences (SPSS) version 23.0 (IBM, New York). Continuous variables with normal distribution were expressed as mean ± standard deviation (range); non-normal variables were reported as median (interquartile range). The statistical significance was determined by the paired t-test for continuous scale variables. P < 0.05 was considered statistically significant.
RESULTS
Patient spectrum
The mean (± SD) age of the patients was 40.3 ± 14.9 years, ranging from 19 to 64 years. There were 12 males and six females [Table 1]. All the patients had radiculopathy. Myelopathy was present in 12 cases. The most common symptom was a backache. In six cases, the lesion was extending beyond one vertebral body level to involve nearly one and a half vertebral body length. It was involving one vertebral body level in all other cases. Only one case had an extraforaminal extension.
| Age (years) (mean+SD [range]) | 40.3±14.9 (19–64) |
|---|---|
| Gender (M: F) | 2:1 |
| Pathology | |
| Neurofibroma/schwannoma | 13 (72.2) |
| Meningioma | 5 (27.8) |
| Levels operated (=n cases) | |
| Cervical | 3 (16.7) |
| Thoracic | 9 (50) |
| Lumbar/lumbosacral | 6 (33.3) |
| Duration of surgery (minutes) | 157±45.3 (90–240) |
| Intraoperative blood loss (mL) | 168.8±78.8 (30–300) |
| Bony exposure | |
| Hemilaminectomy | 13 (72.2) |
| Interlaminar approach | 5 (27.8) |
| Post-operative hospital stays (days) | 4.29±1.4 (2–7) |
| Post-operative complications (=n) | |
| CSF leak | Nil |
| Pseudomeningocele | 1 |
| Bladder dysfunction | 2 |
| Spasticity (increased) | 1 |
| Persistent paresthesia | 3 |
| Weakness | 4 |
| CRPS | 1 |
| Duration of follow-up (months) | 19.3±7.2 (7–36) |
| Nurick’s grade | |
| Pre-operative | 2.6±1.4 (1–5) |
| At latest follow-up | 1.3±1.6 (0–5) (1 [3]) |
| Mean change in Nurick’s grade | 1.29±0.6 (0–3) (P<0.01) |
| ODI score | |
| Pre-operative | 53.3±20.8 (20–80) |
| At latest follow-up | 24.0±22.7 (0–80) (10 [25]) |
| Mean % age change in ODI score | 29.5±13.3 (0–60) (P<0.05) |
Operative findings
Hemilaminectomy was done in 13 patients (n = 13/72.2%). Additional partial adjacent hemilaminectomy for wider exposure was required in six (n = 6/33.3%). The interlaminar corridor was used in five cases (n = 5/38.4%). Total excision was achieved in all the patients. For patients with neurofibroma and meningioma, the respective operative time (163.1 ± 48.3 vs. 134 ± 27.9), blood loss (166 ± 83 vs. 172 ± 65), and hospital stay (4 ± 1 vs. 5 ± 2) were comparable. However, the complications rates were higher in meningioma (n = 3/60%) as compared to neurofibroma (n = 3/23.1%).
Post-operative outcome
One patient had pseudomeningocele formation [Figure 3]. However, there was no CSF leak or wound complication. Most of the post-operative complications (paraparesis or bladder dysfunction) were transient and recovered during follow-up. The patient of cervical neurofibroma had post-operative complex regional pain syndrome. His symptoms are under control with medications. The mean immediate post-operative ODI was 34.0 ± 12.65 (range 20–60) and the mean immediate post-operative Nurick’s grade was 1.7 ± 1.5 (range 0–4). The percentage change in the ODI score and the Nurick’s grade in the post-operative period was statistically significant (paired t-test, P < 0.05).
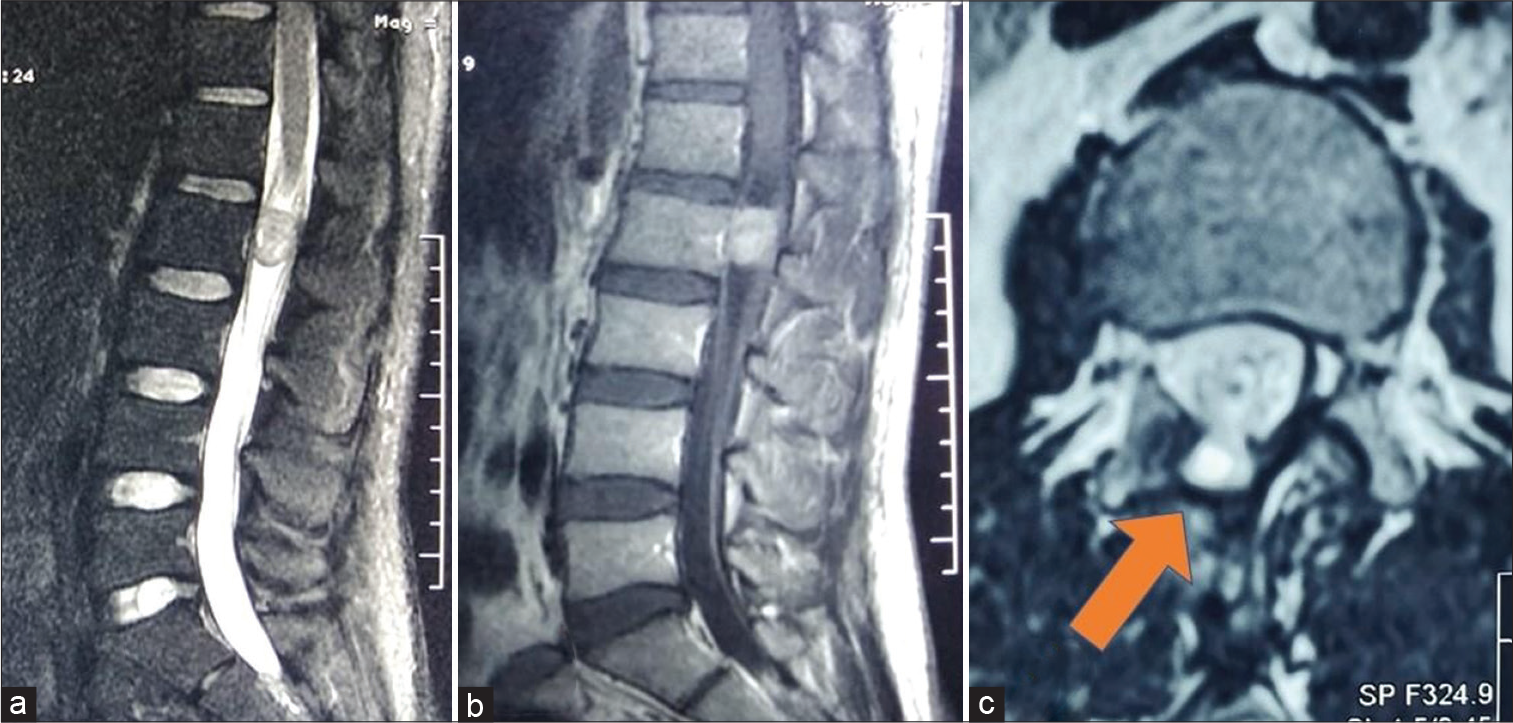
- (a) Pre-operative image – T2 sagittal MRI showing heterogeneous, dominantly isointense IDEM lesion at L2 level. (b) Pre-operative image – T1 sagittal contrast-enhanced MRI showing homogenously contrast-enhancing IDEM at the L2 level. (c) Post-operative image – T2 axial image at L2 vertebral body level showing pseudomeningocele (arrow) herniating through the hemilaminectomy defect. The overlying muscle planes are well maintained.
DISCUSSION
Following endoscopic excision for IDEM, patients have less post-operative pain, earlier mobilization, and less postoperative hospital stay as compared to open surgery.[1] However, if there is a CSF leak or wound bulge, these benefits cannot be realized to their full potential. In this fear, sometimes, the patients are ambulated late. Thus, the importance of watertight dural closure cannot be over-emphasized, especially in minimally invasive spine surgery.
Surgical nuances of endoscopic resection of IDEM using Destandau’s retractor
In minimally invasive techniques, especially while operating for lesion of thoracic spine, wrong level docking is common. It can be avoided by a two-step confirmation of level using the C-arm. First, the localization of the level should be done before draping the patient using a long needle. We used spinal needle of 18 G inserted contralateral to the intended side, at the lamina. Contralateral insertion prevents formation of hematoma in the operative corridor. Second, at the time of docking of the Destandau’s retractor, the dural bulge due to the underlying tumor is usually evident following boney exposure.
In cases of the lower lumbar IDEM, an interlaminar approach can be used without any hemilaminectomy. In thoracic spine, however, hemilaminectomy is usually needed as interlaminar space is hardly present. Moreover, for meningiomas, it is better to have a more extensive exposure, thus hemilaminectomy is appropriate. Neurofibroma might be approached by a pure interlaminar approach as it has a limited attachment. A greater extent of attachment of meningioma also explains the higher incidence of postoperative paraparesis or paraplegia as compared to patients of neurofibroma.
Undercutting of the spinous process gives good exposure of even the contralateral foramina. Thus, allowing a 270° exposure around the spinal cord from unilateral approach. Only the contralateral anterolateral quadrant cannot be reached. However, it does not limit the utility of this technique as the tumors are usually more towards one side and are approached through the same side.
We preferred to open the dura midway between lateral exposure and the midline. This served two benefits. First, the dura toward the midline was retracted with the Destandau’s retractor providing good exposure. Second, being near midline, the artificial dura could be tucked under the spinous process, thus preventing it from getting dislodged.
Existing techniques of dural closure in MIS IDEM excision
CSF leak rate in cases of MIS IDEM excision ranges from 0% to 5% as compared to 0% to 9% in open cases.[11-13] Dhandapani et al.[5] on 34 cases of IDEM excision using either the microendoscopic or the pure endoscopic technique, the authors were able to suture dura in 22 out of 34 cases, only tagging of dura was done in nine cases and they were unable to suture the dura in three cases. One of their patients had a wound bulge which was managed conservatively and it resolved in 2 weeks, while none had a CSF leak. In the study by Wong et al.,[1] the authors analyzed the open versus MIS approach in 45 consecutive patients. The authors closed the dura using running 4–0 Nylon suture. They had a total of four cases of CSF leak (three out of 14 cases in open technique and one out of 24 cases in MIS approach) out of which three cases needed resurgery (two in the open group and one in MIS group). Various methods such as suturing (including the Knot pushing technique),[6-8] tagging of the dura, and applying clips, namely, U clips or titanium clips,[6,9,10] have been described to counter the risk of CSF leak or pseudomeningocele formation. The need for specialized instruments and a specific technique might make the technique less useful for many surgeons.
Efficacy of artificial dura in preventing CSF leak in MIS IDEM excision
In our study, we found the artificial dural layer to be as effective as any other technique described in the literature. None of the patients in our study had any CSF leak or symptomatic pseudomeningocele or any wound-related complication. Due to minimal tissue handling, the dead space in the deeper part of the operative cavity is significantly reduced and as the muscles tend to “fall back to their place” after the endoscope system is removed, the support provided by the overlying muscle helps in further reducing the dead space. Moreover, after the IDEM tumor is removed, the dural edges also tend to fall over inside the thecal sac further reducing the “dural opening.” No special instruments are required, unlike in suturing the dura. We did not use fibrin glue in any of our patients. Fibrin glue is used over the sutured dura in most centers.
Learning curve and operative outcome: Impact of artificial dura
Parihar et al. had reported a series of 18 patients of IDEM excised using endoscope with retractor system.[14] The postoperative complications were mostly reported in the initial seven cases in our series. This represents the early learning curve of the author. However, the operative time in this study was comparable with other authors. This is because of the advantage conferred using artificial dura instead of the tedious technique of dural sutures. For the fear of possible wound-associated complications, Parihar et al. mobilized their initial six patients in whom dural sutures were not taken only after 72 h. In our study, all the patients were mobilized within 24–48 h following surgery with no wound-associated complications.
CONCLUSION
With good efficacy in the prevention of CSF leak from dural rent, cost-effectiveness, and technical ease, dural closure with artificial dura is a favorable technique. It effectively circumvents the steep learning curve of endoscopic IDEM excision.
Declaration of patient consent
Patient’s consent not required as there are no patients in this study.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Comparison of open and minimally invasive surgery for intradural-extramedullary spine tumors. Neurosurg Focus. 2015;39:E11.
- [CrossRef] [PubMed] [Google Scholar]
- A Biomechanical evaluation of graded posterior element removal for treatment of lumbar stenosis: Comparison of a minimally invasive approach with two standard laminectomy techniques. Spine (Phila Pa 1976). 2009;34:17-23.
- [CrossRef] [PubMed] [Google Scholar]
- Biomechanical comparison of traditional and minimally invasive intradural tumor exposures using finite element analysis. Clin Biomech (Bristol Avon). 2009;24:143-7.
- [CrossRef] [PubMed] [Google Scholar]
- Minimally invasive cervical microendoscopic foraminotomy: An initial clinical experience. Neurosurgery. 2002;51:S37-45.
- [CrossRef] [Google Scholar]
- "Microendoscopic" versus “pure endoscopic” surgery for spinal intradural mass lesions: A comparative study and review. Spine J. 2018;18:1592-602.
- [CrossRef] [PubMed] [Google Scholar]
- Minimally invasive surgery for synchronous, same-level lumbar intradural-extramedullary neoplasm and acute disc herniation. Neurosurg Focus. 2014;37:Video16.
- [CrossRef] [PubMed] [Google Scholar]
- A novel minimally invasive technique for spinal cord untethering. Neurosurgery. 2007;60:ONS70-74.
- [CrossRef] [PubMed] [Google Scholar]
- Minimally invasive resection of intraduralextramedullary spinal neoplasms. Neurosurgery. 2006;58:ONS52-8.
- [CrossRef] [PubMed] [Google Scholar]
- Dural closure using the U-clip in minimally invasive spinal tumor resection. J Spinal Disord Tech. 2010;23:486-9.
- [CrossRef] [PubMed] [Google Scholar]
- The use of titanium non-penetrating clips to close the spinal dura. Br J Neurosurg. 2007;21:268-71.
- [CrossRef] [PubMed] [Google Scholar]
- Minimally invasive approach for the resection of spinal neoplasm: Spine (Phila Pa 1976) . 2011;36:E1018-26.
- [CrossRef] [PubMed] [Google Scholar]
- A comparison of mini-open and open approaches for resection of thoracolumbar intradural spinal tumors. J Neurosurg Spine. 2011;14:758-64.
- [CrossRef] [PubMed] [Google Scholar]
- Spinal neurofibromas: A report of 66 cases and a comparison with meningiomas. Neurosurgery. 1986;18:331-4.
- [CrossRef] [PubMed] [Google Scholar]
- Endoscopic management of spinal intradural extramedullary tumors. J Neurol Surg Part Cent Eur Neurosurg. 2016;78:219-26.
- [CrossRef] [PubMed] [Google Scholar]






