Translate this page into:
Reversible dementia in a case of functional gonadotropin-secreting pituitary adenoma: Different shades of gray
*Corresponding author: Dhaval P. Shukla, Department of Neurosurgery, National Institute of Mental Health and Neurosciences, Bengaluru, Karnataka, India. neurodhaval@rediffmail.com
-
Received: ,
Accepted: ,
How to cite this article: Singha S, Mathuranath PS, Shukla DP, Uppar A, Rao S. Reversible dementia in a case of functional gonadotropin-secreting pituitary adenoma: Different shades of gray. J Neurosci Rural Pract. 2024;15:407-9. doi: 10.25259/JNRP_66_2024
Dear Editor,
Functional gonadotropin-secreting pituitary adenomas (FGPAs) secret one or both types of gonadotropins, namely, luteinizing hormone (LH), and follicle-stimulating hormone (FSH), in active biological forms.[1] The FGPA presenting with dementia is extremely rare.[2] In this paper, we report a case of FGPA presenting with dementia, which improved significantly following surgery.
This 58-year-old lady presented with fatigability, apathy, depressive symptoms urinary incontinence, and forgetfulness affecting activities of daily living for one year with behavioral changes. Her Karnofsky Performance Status (KPS) and Eastern Cooperative Oncology Group Performance Status (ECOG PS) were 70 and 2, respectively. Neurological examination revealed a mini-mental state examination score (MMSE) of 22 out of 30, and Addenbrooke’s cognitive examination (ACE III) revealed a total ACE III score of 72 out of 100. Ophthalmological examination revealed bi-temporal hemianopia with the right eye visual acuity of hand movement perceived close to face and left eye visual acuity of 6/60. Magnetic resonance imaging brain revealed a sellar-supra supra-sellar lesion measuring 3.62 cm × 3.50 cm × 4.28 cm in size, Hardy Wilson Grade IIC, Knosp grade 0 [Figure 1]. Her endocrine workup revealed markedly elevated serum LH (>200 mIU/mL), and FSH (>200 mIU/mL) levels with mild elevation of prolactin levels (42.87 ng/mL), and the rest were normal. Hence, the lesion was diagnosed as a gonadotropin-secreting pituitary tumor. She underwent an endoscopic trans-nasal trans-sphenoidal approach and near-total decompression of the lesion. The final diagnosis was gonadotroph-adenoma [Figure 1]. The patient had an uneventful post-operative recovery, and a follow-up scan at six months showed gross total resection of the lesion [Figure 1]. Post-operative LH (0.15 mIU/mL) and FSH (0.66 mIU/mL) levels were normalized with significant neurocognitive improvement. At the three-month follow-up, her MMSE and ACE III scores were 28/30 and 93/100, respectively. Her vision has improved in both eyes (right eye 6/60 and left eye 6/6) with significant improvement in visual fields. Overall, her functional status (KPS 100, ECOG PS 0), cognitive and behavioral symptoms, and urinary incontinence improved significantly.
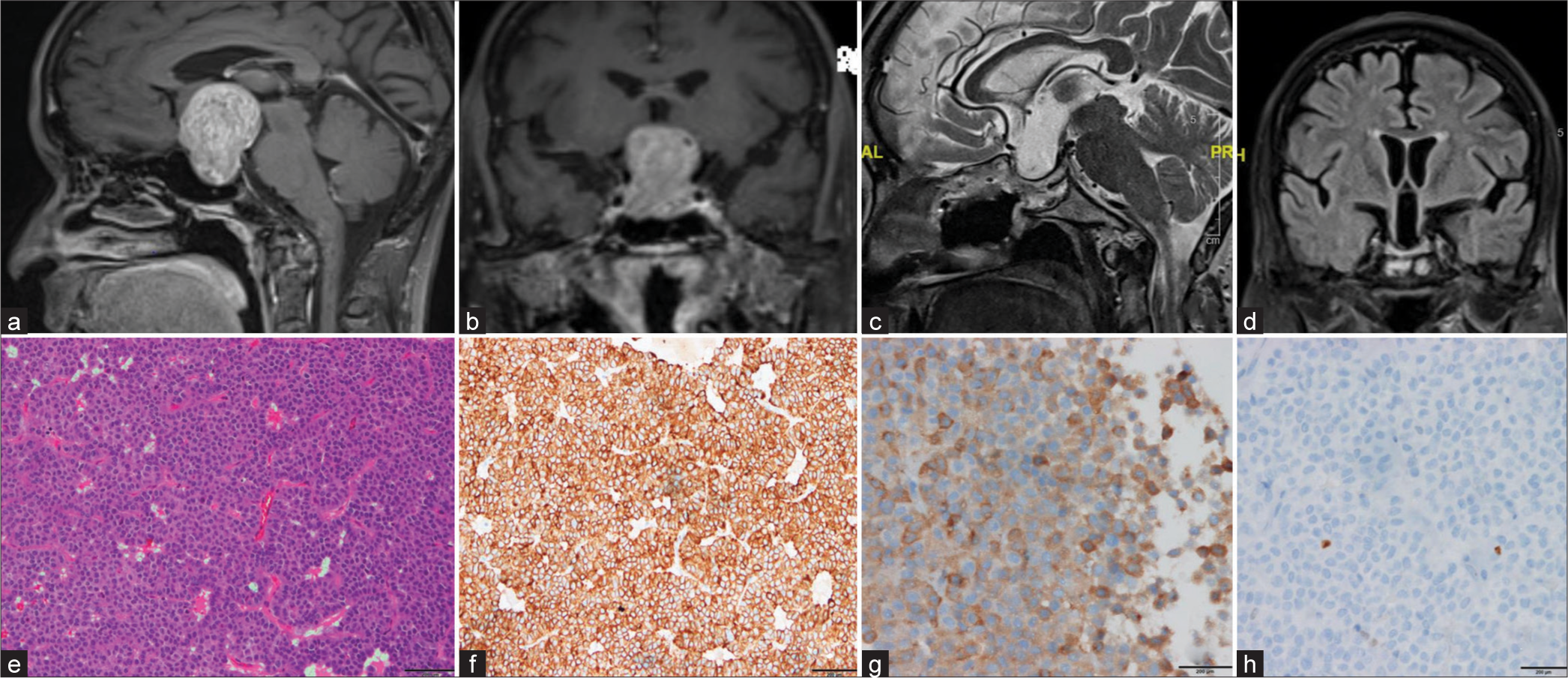
- (a and b) Pre-operative magnetic resonance imaging (MRI) brain (T1 Contrast-weighted sequences) shows a heterogeneously enhancing sellar-supra supra-sellar lesion measuring 3.62 cm × 3.50 cm × 4.28 cm in size, anteriorly draping the anterior communicating artery, bilateral A1 segment of an anterior cerebral artery, laterally, being limited by bilateral internal carotid artery bifurcation, superiorly extending into the cavity of the third ventricle reaching up to the foramen Monro without obstructing it. (c and d) Post-operative MRI shows an operative cavity occupied by cerebrospinal fluid in the sellar-suprasellar region without any residual/recurrent disease. (e) Photomicrograph showing histopathological characteristics of the pituitary adenoma; the hematoxylin and eosin stains show that the lesion is composed of polygonal to round cells arranged as sheets, papillae, and nests separated by fibrovascular septae with cells being mildly pleomorphic with a round nucleus, stippled chromatin, and moderate acidophilic to chromophobic cytoplasm with occasional mitoses. (f and g) Cells are immunopositive for cytokeratin and Luteinizing hormone respectively. (h) The proliferation index (Ki 67) is 2%.
Memory and behavioral disturbances in pituitary adenomas can be due to underlying hypothyroidism, adrenal insufficiency, lobar extension (frontal and temporal), and normal pressure hydrocephalus (NPH).[2] In our case, there were no radiological or clinical features of NPH, and the classical triad of NPH was incomplete due to the absence of gait disturbance. In this case, the possible mechanism of urinary incontinence was disinhibition of the pontine micturition center by frontal lobe dysfunction caused due to the mass effect of the tumor due to forebrain excitatory pathway upregulation and downregulation of forebrain tonic inhibitory pathways. Various studies propose that there is upregulation of glutamatergic (NMDA) and D2 dopaminergic excitatory pathways in the absence of suprapontine inhibition giving rise to an overactive bladder causing social incontinence.[3] Hence, when the offending lesion was removed, there was a complete reversal of urinary incontinence in this patient. The clinical and radiological examination and hormonal evaluation ruled out other causes of dementia in pituitary adenoma (central hypothyroidism, adrenal insufficiency, NPH, increased intracranial pressure, and frontal lobar involvement). The role of pituitary gonadotropins and ovarian steroids has long been discussed in postmenopausal cognitive decline and the pathogenesis of Alzheimer’s disease (AD).[4] The secretion of FSH and LH is strongly regulated by complex feedback loops of the hypothalamic-pituitary-ovarian axis. In postmenopausal women, with the reduction in estrogen levels, there is a loss of negative feedback on gonadotropins causing higher circulating levels of LH and FSH. Key factors in the development of cognitive decline in AD are the deposition of amyloid-b (Ab) in and around the cerebral neurons causing neuronal death and cerebral atrophy.[4] Females have a more substantial accumulation of amyloid senile plaques and neurofibrillary tangles, the pathological hallmarks of AD as compared to their male counterparts. Now, it is found that LH modulates the processing of amyloid-b protein precursor and generation of Ab in vitro, and various gonadotropin-releasing hormone agonists also reduce Ab levels in mouse brain.[4] To summarize, an increased level of gonadotropins can cause neurodegeneration in an aging brain. The presence of the Apo-lipoprotein e4 allele plays a key role in the development of AD. In our case, there was subsequent normalization of levels of gonadotropins following surgery, which resulted in a reversal of the symptoms and near-complete recovery in her clinical status.
This is the second case report of gonadotroph-adenoma in published literature, which has presented with dementia and improved completely following surgical removal of the tumor.[5]
Ethical approval
The Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using the AI.
Financial support and sponsorship
Nil.
References
- Pituitary tumors: Epidemiology and clinical presentation spectrum. Hormones (Athens). 2020;19:145-55.
- [CrossRef] [PubMed] [Google Scholar]
- Dementia and low-pressure hydrocephalus in a patient with pituitary adenoma. J Neurol Sci. 1973;19:341-9.
- [CrossRef] [PubMed] [Google Scholar]
- Glutamatergic and dopaminergic contributions to rat bladder hyperactivity after cerebral artery occlusion. Am J Physiol. 1999;276:R935-42.
- [CrossRef] [PubMed] [Google Scholar]
- Gonadotropins and cognition in older women. J Alzheimers Dis. 2008;13:267-74.
- [CrossRef] [PubMed] [Google Scholar]
- Effectiveness of endoscopic transsphenoidal surgery for gonadotroph adenoma mimicking dementia: A case report. Intractable Rare Dis Res. 2019;8:217-20.
- [CrossRef] [PubMed] [Google Scholar]





