Translate this page into:
Pilocytic Midbrain Astrocytoma Presenting with Fresh Bleed after Twenty-one-years Survival Following First Surgery: A Unique Case of Longest Brainstem Glioma Survival
Address for correspondence: Dr. Guru Dutta Satyarthee, Department of Neurosurgery, Neurosciences Centre, All India Institute of Medical Sciences, New Delhi, India. E-mail: duttaguru2002@yahoo.com
This is an open access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 3.0 License, which allows others to remix, tweak, and build upon the work non-commercially, as long as the author is credited and the new creations are licensed under the identical terms.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Brainstem glioma usually carries a poor prognosis and prolonged survival is very infrequent. In a detailed Pubmed, Medline search for prolonged survival, authors could got a longest survival only up to seventeen years, reported by Umehara et al, who was subjected to gamma knife therapy and got symptomatic, MRI brain reveled large tumor growth during pregnancy necessitating emergency surgery and histopathological diagnosis was pilocytic astrocytoma. Authors report an interesting case of midbrain glioma diagnosed 21 years back, who underwent gross resection in the year 1993, histopathology was pilocytic astrocytoma, WHO grade I, and received gamma knife surgery for residual subsequently and he presented with sudden onset left sided hemiplegia on the current admission. The cranial MRI imaging revealed an infarct involving right hemi midbrain, contrast MRI brain revealed no residual glioma. To the best knowledge of authors such prolonged survival is not reported with a case of brainstem glioma survived twenty- one years with non residual tumor on the last imaging study represents first case of its kind in the western literature and probably developed hemiplegia due to bleed, highlighting bleed as delayed complication following gamma knife therapy for cranial tumors
Keywords
Brainstem glioma
midbrain infarct
prolonged survival
surgery
GK therapy
INTRODUCTION
Brainstem glioma usually carries a poor prognosis in children. Favorable prognostic factors of brainstem glioma include the presence of neurofibromatosis, relatively younger age at diagnosis, exophytic components, focal lesion and calcification on computed tomography, histopathology grades. However, prolonged survival is very infrequent.[12] Authors report an extremely uncommon case of brainstem glioma, which underwent surgery 21-years back for pilocytic astrocytoma of the midbrain and also received gamma knife therapy. He presented with hemiplegia due to bleed in hemi-midbrain possibly due to gamma knife-induced arteriopathy.
A 34-year-old male presented with sudden-onset hemiplegia and altered sensorium. Important history included left temporal craniotomy and microsurgical decompression of a focal midbrain mass lesion, pilocytic astrocytoma, WHO Grade I in the year 1993. He also received radiotherapy 3 months following initial surgery. In 1994, he also underwent cerebrospinal fluid (CSF) diversion surgery due to associated hydrocephalus, and medium pressure ventriculoperitoneal (VP) shunt placement was carried out. In 1995, he underwent re-exploration surgery and removal of residual mass lesion. During the follow-up period after the second surgery, he was doing well, except a small recurrent lesion was observed in magnetic resonance imaging (MRI), for which he received gamma knife therapy in 2002.
At present admission, he presented with sudden-onset hemiplegia and altered sensorium. Evaluation on admission revealed a Glasgow coma scale (GCS) of E2V2M5 with left-sided facial nerve paresis of lower motor neuron type along with left-sided hemiplegia. MRI of the brain on admission revealed an infarct in the hemi-midbrain showing hypointense signal on the T1-weighted image. It was showing restriction of diffusion in the diffusion-weighted images [Figure 1], hypointense signal on T2-weighted image [Figures 2 and 3]. He was electively ventilated for 1 week, and tracheostomy was performed for pulmonary toileting. He was gradually weaned off from the ventilator and discharged with a GCS of E4VtM6 and walking with support accepting oral feed and continuing daily activity of living with support.
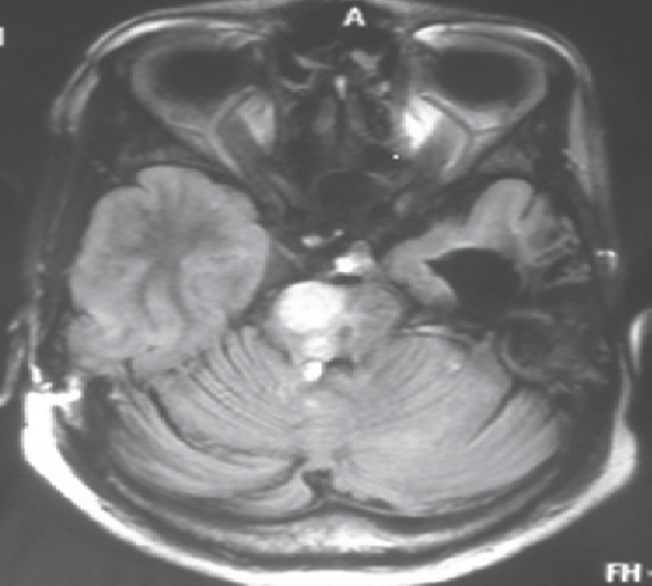
- Magnetic resonance imaging of the brain of 34-year-old male, diffusion-weighted axial section image showing hypointense lesion in the right half of the midbrain
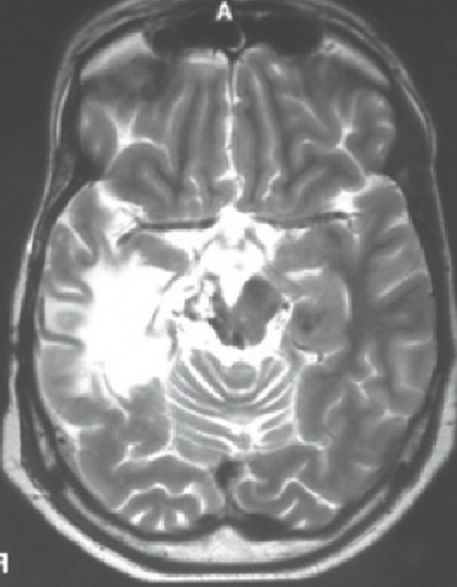
- The cranial magnetic resonance imaging, axial section, T2-weighted image showing hypointense lesion in the right half of the midbrain
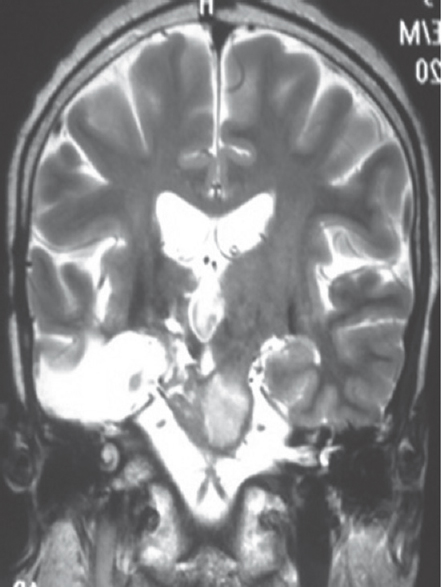
- Magnetic resonance imaging of the brain, coronal section T2-weighted image showing lesion in the right half of the midbrain with hypointense signal intensity
Brain stem glioma is one of the common tumors in pediatric age group. It originates from usually glial cells.[1] Depending on histopathology, it can be categorized as UN WHO Grade I, II, III, or IV.[234] Brainstem gliomas can further also be categorized into following groups, on the basis of neuroimaging features, i.e., diffuse, exophytic-focal, intrinsic-focal, cervicomedullary, and types based on radiological features.[4]
Common differential of brainstem mass is demyelinating brainstem pathologies, tuberculosis, and inflammatory. Usually, diagnosis is finally based on histopathological features analyzed in relation to the imaging study's findings.[56]
Various treatment modalities of brain stem glioma are highly variable and including forms a spectrum of approaches including, stereotactic biopsy, resection alone or combined with adjuvant therapy for suboptimally resected mass, or supervised observation with serial imaging study or adjunctive CSF diversion therapy like VP shunt surgery followed by primary radiation therapy, chemotherapy in different combinations depending on respectability of lesion, clinical status of patient, experience of surgical team, availability of adjuvant therapy facility.
Surgery is usually advocated in focal, exophytic, and cervicomedullary lesions or midbrain.[78910] Stereotactic biopsy of brainstem mass is other minimally invasive surgical option to obtain tissue for final histopathological and immunohistochemistry graqding and typing Selvapandian et al. observed Grade II astrocytoma being the most common variety in their series of 92 cases, who underwent stereotactic biopsy.[1] However, in addition to primary surgery, many cases may need VP shunt surgery to treat hydrocephalus, which develops secondary to obstruction in CSF flow pathway due to the growth of brain stem mass.
Although brainstem gliomas carry poor prognosis and shorter life expectancy, few patients may have prolonged survival.[1112131415] Interestingly, our case is unique as he survived for 21 years after histopathological confirmation after diagnosed as brainstem glioma. Prognostic factors affecting survival in the brainstem glioma include age at diagnosis, histopathological type and WHO grade, extent of surgical resection, adjuvant therapy, focal or diffuse lesion, requirement for VP shunt, and associated comorbid illness.[1415] Umehara et al. reported a case of pilocytic astrocytoma who received gamma knife therapy 17-years back developed rapid tumor growth during pregnancy necessitating emergency surgery and histopathological diagnosis was pilocytic astrocytoma. However, only partial resection was possible as it was infiltrating the brainstem parenchyma.[16]
However, our case presented at the current admission with infarct of hemi-midbrain, which can be attributed to bleed in residual tumor or due to an effect of Gamma knife therapy. Gamma knife therapy is known to cause arteriopathy not only in the tumor bed but also in the adjoining regions of the brainstem.[121718] However, it remains a speculation, if it was bleeding due to malignant transformation which can occur de novo by conversion into higher grade with neoangiogenesis leading to tumor bleed.[161920] However, a biopsy would have clearly demonstrated the exact nature of cause and histopathological diagnosis. However, more evidence lies in favor of radiation-induced arteriopathy following gamma knife therapy as there is a uniform restriction of diffusion in diffusion-weighted MRI study images.
Brainstem glioma usually carries a bad prognosis and prolonged survival is very infrequent. In a detailed PubMed, Medline search for prolonged survival, authors could get the longest survival only up to 17-years, reported by Umehara et al., who was subjected to gamma knife therapy without preoperative histopathological confirmation.[16] She, later on, got symptomatic, MRI brain revealed large tumor growth during pregnancy necessitating emergency surgery, and histopathological diagnosis was pilocytic astrocytoma was only made 17 years after the first clinical presentation.
A total of 58 cases of brainstem glioma were managed by authors.[2] Of these cases, the current case has the longest survival. Our case is interesting as he underwent first surgery about 21 years back, for brain stem glioma, in the year 1993, underwent surgical re-exploration for recurrence, and also received gamma knife therapy. Following gamma knife therapy, he was doing well with good physical and cognitive performance and independently managing automotive spare parts shop and he presented with sudden-onset left-sided hemiplegia on the current admission. The cranial MRI imaging revealed an infarct involving right hemi-midbrain, contrast MRI brain revealed no residual glioma. To the best of our knowledge, such prolonged survival is not reported with a case of histopathologically proven pilocytic astrocytoma of brain stem, who survived 21 years with nonresidual tumor on the last imaging study and probably developed hemiplegia due to bleed, highlighting bleed as delayed complication following gamma knife therapy for cranial tumors and represents first case of its kind reported in the literature.
Although brainstem glioma carries a poor prognosis, a few cases may report prolonged survival, highlighting the hope for prolonged disease-free survival as in the current case. Hence, these cases may exhibit better response with multimodalities treatment with close supervision. As our cases needed gamma knife surgery as well as CSF diversion surgery for residual lesion and progressive obstructive hydrocephalus, respectively, every case of brainstem glioma developing new neurological deficit during the follow-up period warrants at least MRI brain as investigation modality of choice to exclude not only tumor recurrence but also to exclude infarct and delayed radionecrosis, and these may be representing sequelae of radiation therapy.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- Brainstem glioma: Comparative study of clinico-radiological presentation, pathology and outcome in children and adults. Acta Neurochir (Wien). 1999;141:721-6.
- [Google Scholar]
- Brainstem gliomas:Surgical indications and technical considerations in a series of 58 cases. Br J Neurosurg. 2014;28:220-5.
- [Google Scholar]
- The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol. 2007;114:97-109.
- [Google Scholar]
- Brainstem tumors. In: Choux M, Di Rocco C, Hockley A, eds. Pediatric Neurosurgery. New York: Churchill Livingstone; 2000. p. :471-91.
- [Google Scholar]
- The efficacy of a biopsy of intrinsic brainstem lesions for decision making of the treatments. Childs Nerv Syst. 2013;29:833-7.
- [Google Scholar]
- Clinical outcomes and natural history of pediatric brainstem tumors: With 33 cases follow-ups. Neurosurg Rev. 2013;36:311-9.
- [Google Scholar]
- Surgical management of brain-stem tumors in children: Results and statistical analysis of 75 cases. J Neurosurg. 1993;79:845-52.
- [Google Scholar]
- Juvenile pilocytic astrocytoma of the brainstem in children. J Neurosurg. 2004;101(1 Suppl):1-6.
- [Google Scholar]
- Phase I trial of imatinib in children with newly diagnosed brainstem and recurrent malignant gliomas: A pediatric brain tumor consortium report. Neuro Oncol. 2007;9:145-60.
- [Google Scholar]
- Correlation among magnetic resonance imaging findings, prognostic factors for survival, and histological diagnosis of intrinsic brainstem lesions in children. J Neurosurg Pediatr. 2011;8:539-43.
- [Google Scholar]
- Young age may predict a better outcome for children with diffuse pontine glioma. Cancer. 2008;113:566-72.
- [Google Scholar]
- A case of pilocytic astrocytoma requiring tumor resection during pregnancy. Mol Clin Oncol. 2016;4:567-70.
- [Google Scholar]
- Radiological response and histological changes in malignant astrocytic tumors after stereotactic radiosurgery. Brain Tumor Pathol. 2002;19:83-92.
- [Google Scholar]
- Can radiation induce pituitary tumors? Giant prolactinoma after radiation exposure. J Clin Neurosci. 2008;15:1287-8.
- [Google Scholar]
- Isolated brain metastasis from malignant melanoma of choroid seven years following enucleation. Neurol India. 2009;57:92-4.
- [Google Scholar]






