Translate this page into:
Guillain-Barre syndrome with posterior reversible encephalopathy syndrome
Address for correspondence: Dr. Basavaraj Banakar, Department of Neurology, M G Hospital, Near Jalori Gate, Jodhpur, Rajasthan, India. E-mail: drbanakar2006@yahoo.co.in
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Posterior reversible encephalopathy syndrome (PRES) is a clinicoradiologic entity commonly associated with eclampsia, septicemia, chemotherapeutic drugs etc. Concurrent occurrence of Guillain-Barre syndrome (GBS) with PRES is a rare entity. Dysautonomia is a proposed mechanism for such occurrence. Here we present a non-diabetic, non-hypertensive 63-year-old male patient, who came with acute onset flaccid quadriparesis, developing generalized seizures, altered sensorium and raised blood pressure on fifth day of illness. Magnetic resonance imaging (MRI) of brain showed altered signal intensities involving the parieto-occipital areas suggestive of posterior reversible encephalopathy. Cerebrospinal fluid analysis showed albuminocytological dissociation, nerve conduction studies revealed demyelinating type of polyneuropathy. The patient was treated with antihypertensives and antiepileptics. After resolution of the encephalopathy, intravenous immunoglobulin (IVIg) was given. The patient recovered gradually over few months. Our case concludes GBS as independent risk factor, for PRES may be secondary to dysautonomia and physicians should be aware of such rare coexistence so that early treatment can be done to reduce the mortality and morbidity.
Keywords
Guillain-Barre syndrome
intravenous immunoglobulin
posterior reversible encephalopathy syndrome
Introduction
Posterior reversible encephalopathy syndrome (PRES) is a clinicoradiologic entity which manifests with headache, seizure, altered sensorium. It is being diagnosed more often with varied etiologies after the advent of MRI. Eclampsia, septicemia, chemotherapeutic drugs are some of the common causes. Though Guillain-Barre syndrome (GBS) with PRES following the intravenous immunoglobulin therapy (IVIg) had been mentioned, however concurrent occurrences of GBS with PRES are rarely reported. Here we present one such case.
Case Report
A 63-year-old non-diabetic and non-hypertensive male patient came with complaints of difficulty in walking of five days, inability to do activities of daily living with upper limbs of four days, dysarthria, dysphagia of two days duration, for which he was admitted in the local hospital. On the day of admission he had four generalized tonic-clonic seizures over the interval of 12 h. After the last seizure he did not regain consciousness and was referred to us. There was no history suggestive of sensory and bladder involvement. History was negative for fever, headache, vomiting, rashes, chronic drug intake or addictions.
On examination, for first three days the patient was stuporous with high blood pressure of 192/94 mmhg, flaccid limbs and bilateral plantar extensor. On fourth day, the patient regained consciousness and examination revealed bilateral lower motor neuron type of facial and bulbar weakness, hypotonic limbs with power of 1/5 in muscles of the shoulder, arm, hip, thigh and 2/5 in forearm and leg (according to MRC grading) along with the weakness of small muscles of the hand. Deep tendon reflexes were absent without sensory loss.
On investigation hemogram, serology, routine biochemistry, chest X-ray, electrocardiogram, echocardiography and abdominal sonography were normal and did not reveal any cause for hypertension. Cerebrospinal fluid analysis showed albuminocytological dissociation with 4-lympocytes, 176 mg/dl-of protein and normal sugar. No organisms were isolated on staining/culture. MRI brain showed bilateral cortical and subcortical altered signal intensities involving the parieto-occipital areas. They were hypointense on-T1W images, hyperintense on-T2W and FLAIR images with no diffusion restriction suggestive of PRES [Figures 1 and 2]. Nerve conduction studies showed prolonged latencies, reduced conduction velocity and absent f waves with reduction in the amplitude of compound muscle action potentials with normal sensory potentials suggestive of demyelinating type neuropathy according to HO et al. criteria.
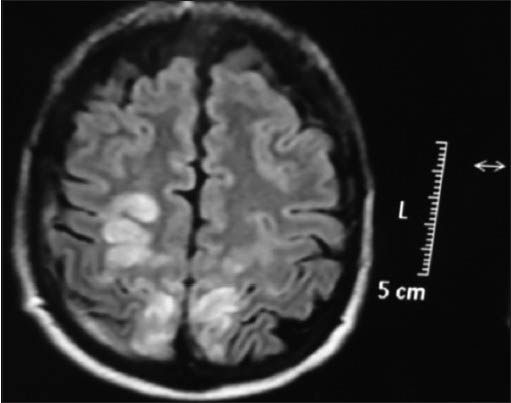
- MRI brain axial section flair image showing bilateral parietoccipital hyperintense signals
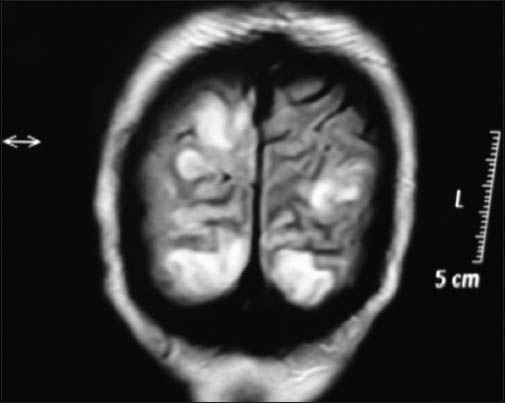
- MRI brain coronal section flair image showing bilateral parietoccipital hyperintense signals
The patient was treated with antihypertensives and antiepileptics initially to control his blood pressure and seizures. After resolution of encephalopathy, IVIg was given and his weakness improved over two months. Now after 6-months, the patient is ambulatory and is independent in his daily activities.
Discussion
Our case is one of the rare presentation of the GBS. GBS is polyradiculoneuropathy involving peripheral nervous system without affecting the sensorium. Sometimes severe respiratory paralysis may lead to hypoxic brain injury leading to altered sensorium/seizures. In our case, the patient did not have respiratory involvement but still had cluster of seizures with loss of consciousness and imaging was classical of PRES. It is the rare combination of GBS with PRES before initiation of therapy with IVIg may be secondary to dysautonomia.
The review of literature shows that the incidence of GBS is 0.75-2 cases per-100000 and the incidence of PRES is even lower than that.[1] Therefore, the coincidence of both GBS and PRES is even rarer. There are few case reports of GBS developing PRES, many of them follow after IVIg therapy.[2] But, PRES developing prior to IVIg therapy are very few.[3] With our case report we want to create awareness among our physicians of such rare coexistence.
PRES is a clinicoradiologic entity described by Hinchey et al.[4] in 1996. This condition has been designated by a variety of names but PRES is now the accepted term. PRES can manifest with headache, nausea/vomiting, seizures, altered sensorium, visual abnormalities, and focal neurological deficits. The cerebral imaging abnormalities are often symmetric and predominate in the posterior white matter. Recognition of PRES has evolved with increasing availability of MRI. PRES is typically reversible once the cause is removed.
PRES can develop in association with a variety of conditions. Common causes[5] include preeclampsia/eclampsia, septicemia, acute hypertension, exposure to chemotherapeutic drugs and autoimmune disorders like systemic lupus erythematosus, systemic sclerosis, polyarteritis nodosa, Wegener's granulomatosis etc. Rarer causes are Guillain-Barre syndrome, hypomagnesemia, hypercalcemia, tumor-lysis syndrome, porphyria, pheochromocytoma, and Cushing syndrome.
However regardless of the underlying cause, the main abnormality is cerebral vasogenic edema, the pathogenesis of which is still under debate.[5] The two main hypotheses contradict each other. One involves impaired cerebral autoregulation responsible for an increase in cerebral blood flow secondary to acute raise in the blood pressure seen in conditions like preeclampsia, eclampsia and autonomic dysfunction, as in our case. Whereas the other involves endothelial dysfunction with cerebral hypoperfusion.[5] This hypoperfusion hypothesis may be most relevant to cases of PRES associated with cytotoxic therapy. Under both hypotheses, the result of the cerebral blood perfusion abnormalities is blood brain barrier dysfunction with cerebral vasogenic edema. The treatment strategy includes general measures, antihypertensives[3] with correction of the underlying cause of PRES. Prognosis is good with early diagnosis and prompt treatment. Permanent neurological deficits can be there but are rare depending on the severity of illness.
Proposed mechanisms leading to PRES in patients of GBS are dysautonomia leading to acute raise in the blood pressure, activation of sympathetic nervous system secondary to pain,[6] IVIG therapy and renal decompensation. In our patient, autonomic dysfunction might explain the cause. PRES can follow or precede the weakness in the patients of GBS, which followed in our case. There are studies to prove that elevated urinary catecholamines[7] correlate with clinical dysautonomia in GBS and can be used as a biological marker, which was not done in our case. Management is by prompt reduction of blood pressure and general measures. There are no consensus[6] between IVIG and plasmapheresis in the patients of GBS developing PRES prior to therapy. We treated the patient with IVIG after reduction of blood pressure.
Conclusion
Our case supports the fact that GBS can be considered as an independent risk factor of PRES secondary to dysautonomia. Failure to recognize or delay the treatment can lead to cerebral infarction or even death. These patients can be treated with IVIG or plasmapheresis. Physicians should be aware of such rare coexistence so that early treatment can reduce the mortality and morbidity.
Acknowledgement
Dr. Bharat Bhushan, DM Neuroresident, Department of Neurology, Dr. S N Medical College, Jodhpur Dr. Gaurav Kasundra, DM Neuroresident, Department of Neurology, Dr. S N Medical College, Jodhpur.
Source of Support: Nil.
Conflict of Interest: None declared.
References
- Posterior reversible encephalopathy syndrome (PRES) in critically ill obstetric patients. Intensive Care Med. 2003;29:2323-6.
- [Google Scholar]
- Neurological complications of intravenous immunoglobulin (IVIg) therapy: An illustrative case of acute encephalopathy following IVIg therapy and a review of literature. Acta Neurol Belg. 1998;98:347-51.
- [Google Scholar]
- Posterior reversible encephalopathy syndrome as a revealing manifestation of Guillain-Barré syndrome. Rev Neurol (Paris). 2012;168:283-6.
- [Google Scholar]
- A reversible posterior leukoencephalopathy syndrome. N Engl J Med. 1996;334:494-500.
- [Google Scholar]
- Understanding posterior reversible encephalopathy syndrome. Annual update in intensive care and emergency medicine 2011. New York: Springer Berlin Heidelberg; 2011. p. :631-53.
- [Google Scholar]
- Posterior reversible encephalopathy associated with the Dysautonomia of Guillain-Barré syndrome. Neurol Bull. 2009;1:7-10.
- [Google Scholar]
- Estimatio of plasma and urinary catecholamines in Guillain-Barre syndrome. Jpn J Med. 1985;24:24-9.
- [Google Scholar]






