Translate this page into:
Expanding the spectrum on brain-heart interactions in cerebellopontine angle surgeries
*Corresponding author: Rohini M. Surve, Department of Neuroanesthesia and Neurocritical Care, National Institute of Mental Health and Neurosciences, Bengaluru, Karnataka, India. rohinigondhule@gmail.com
-
Received: ,
Accepted: ,
How to cite this article: Sharma P, Krishnaswami S, Shah K, Surve RM. Expanding the spectrum on brain-heart interactions in cerebellopontine angle surgeries. J Neurosci Rural Pract. 2024;15:384-6. doi: 10.25259/JNRP_21_2024
Abstract
Lesions at the cerebellopontine angle (CP angle) are associated with various brain-heart interactions, which can include those from stimulation of the fifth cranial nerve along the scalp incision in a retrosigmoid suboccipital surgical approach. A 27-year-old male patient with recently diagnosed hypertension (on calcium channel blocker) underwent left CP angle lesion decompression. Transient episodes of bradycardia, hypotension, and bradypnea were observed from the skin incision onward, exacerbated during tumor manipulation. Most episodes subsided with cessation of the surgical stimulus while some required intervention. Postoperatively, blood pressure decreased below the pre-operative levels. Thus, trigeminocardiac reflex can occur as early as the skin incision even in a retrosigmoid approach due to stimulation of the mandibular division, when specific risk factors exist. Such episodes may serve as early warning signs for subsequent intraoperative occurrences. Brainstem compression can be a possible etiology of hypertension in young patients. It underscores the importance of considering brain-heart interactions in surgical interventions involving the CP angle.
Keywords
Trigeminocardiac reflex
Cerebellopontine angle
Apnea
Hypertension
Brainstem
INTRODUCTION
The trigeminocardiac reflex (TCR) is characterized by abrupt initiation of dysrhythmias (bradycardia), hypotension, bradypnea or apnea, and gut hypermotility following stimulation of sensory branches of the trigeminal nerve.[1] Lesions at the cerebellopontine angle (CP angle) can be associated with various brain-heart interactions.
A young adult with a CP angle lesion, recently diagnosed hypertensive (unknown etiology), experienced multiple episodes of the transient hemodynamic fluctuations which were first observed during the skin incision of the retrosigmoid surgical approach. As muscle relaxants were withheld for cranial nerve monitoring, we observed episodes of apnea under deep propofol sedation along with the expected hemodynamic changes. The patient’s blood pressure (BP) reduced postoperatively to below the pre-operative levels following tumor decompression. Consent for publication was obtained from the patient obviating Institute Ethics Committee approval.
CASE REPORT
A 27-year-old male, recently diagnosed hypertensive on Cilnidipine for the past two months, presented with a heterogeneous lesion in the CP angle with mass effect on the pons, medulla, and fourth ventricle and left cerebellar hemisphere [Figure 1]. Surgical decompression of the tumor was planned using a left retrosigmoid lateral suboccipital approach under general anesthesia (GA), with intraoperative monitoring of the cranial nerves. This case was previously cancelled twice to evaluate the cause of hypertension in an otherwise young adult.
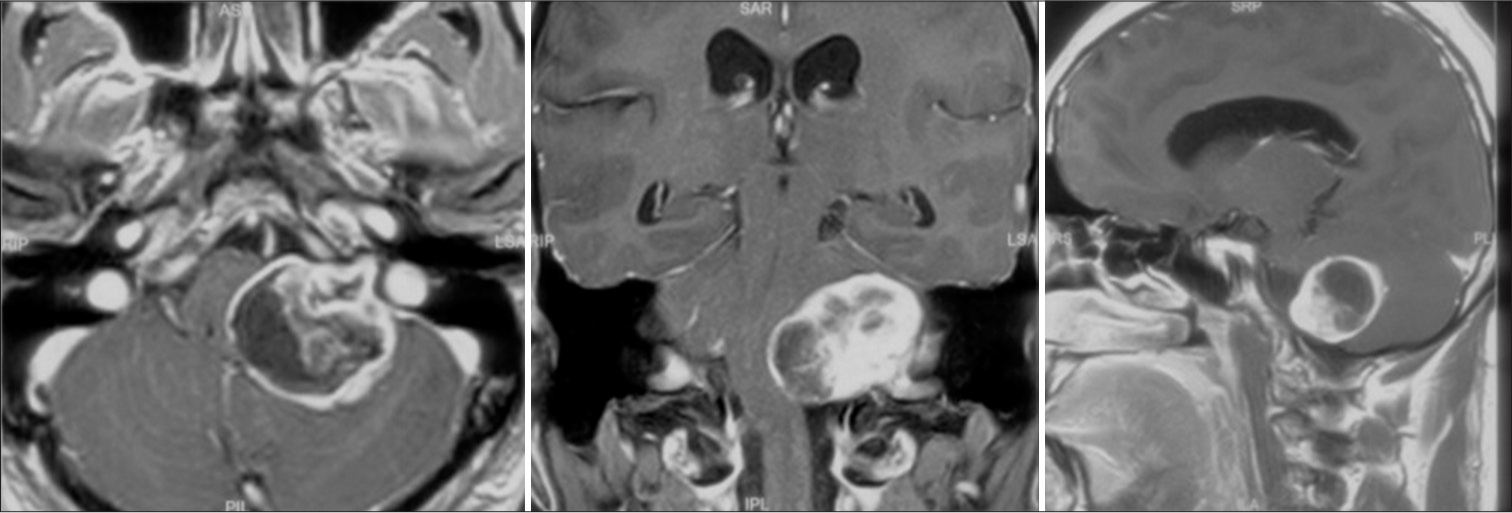
- Post-contrast T1-weighted image in all three planes showing a well-defined extra-axial heterogeneous lesion in the left cerebellopontine angle with extension into the internal auditory canal, causing expansion of the same and significant compression of the pons, medulla, fourth ventricle leading to upstream hydrocephalus and left cerebellar hemisphere.
GA was induced using intravenous fentanyl (2 mcg/kg), thiopentone (5 mg/kg), and rocuronium (1 mg/kg). The patient’s BP pre-induction was 170/95 mmHg (similar to ward BP values), with a heart rate (HR) of 118/min. On making the skin incision [Figure 2] with the patient in the right lateral position, there was a sudden and transient decrease in HR and BP to 20/min and 80/40 mmHg, respectively. The patient’s surgical plethysmographic index was 40 and the bispectral index value 40–50, and capnography and saturation were stable. The surgical team was promptly informed, and the episode settled with the cessation of the surgical stimulus.
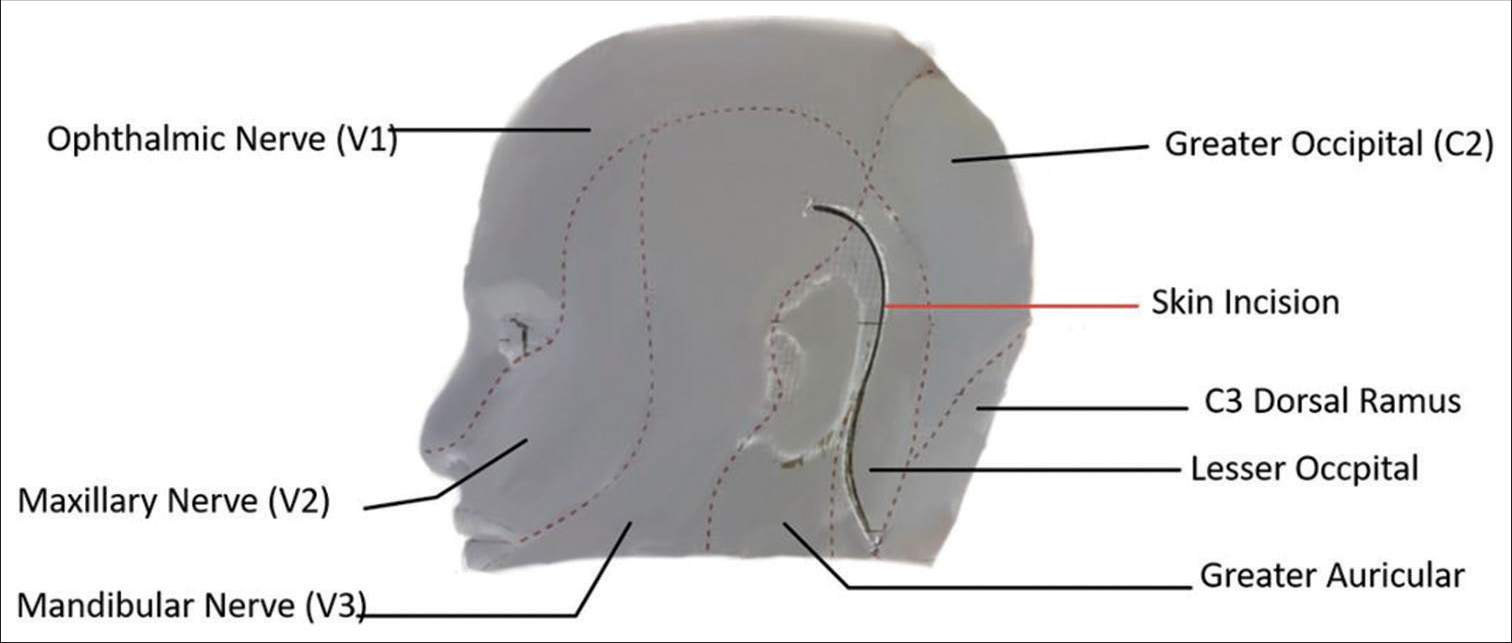
- Surface reconstruction of the scar (red line) with respect to various dermatomes of head based on postoperative magnetic resonance imaging of the patient.
As cranial nerve monitoring was planned, propofol infusion was initiated using a target-controlled infusion (TCI) with an effect site concentration of 2.5–3 mcg/mL and fentanyl (without relaxants). Subsequently, ventilation was changed from volume-controlled to pressure support Pro (PSVPro) mode due to patient triggering. The craniotomy was completed successfully, but while resecting around the tumor adherent to the brainstem, several episodes of simultaneous bradypnea, hypotension, and bradycardia were observed and communicated to the surgical team. Most episodes resolved spontaneously on release of the surgical stimulus (temporal association and not related to blood loss or additional bolus of anesthetic agents), while a few required interventions. The episodes of bradypnea progressed to apnea (Ventilator settings: Pressure support of 15 cm H2O, positive end-expiratory pressure of 5 cm H2O, with a back-up setting of Pinspired 15 cm H2O, and respiratory rate 14/min). The patient was switched back to the controlled mode of ventilation due to frequent switch to backup mode during these episodes. Hypotensive episodes were managed by administering aliquots of mephentermine. Post-tumor decompression, no further changes were observed in the respiration or hemodynamics. The patient was successfully extubated and remained hemodynamically and neurologically stable; his BP reduced and remained lower than the pre-operative levels till discharge.
DISCUSSION
The TCR is a well-recognized phenomenon and can be triggered by the stimulation of any branch of the fifth cranial nerve up to the brainstem centers and by compression of the Gasserian ganglion. The incidence is 11% when resecting around the trigeminal nerve at the brainstem.[2] Although the phenomenon is well established and reported in the literature, it cannot be completely prevented but only anticipated. Predisposing factors (manipulation of the central part of the trigeminal nerve, hypoxia, and hypercapnia, the light plane of anesthesia with strength of stimulus, and potent narcotics and anti-hypertensive agents)[3] for the occurrence of the reflex have been studied but despite that the TCR may be encountered intraoperatively without warning.
There are reports of the occurrence of TCR after skin incision for parietal lesions, and during subgaleal drain placement at closure,[4,5] but the occurrence of TCR at the time of skin incision in CP angle surgeries using the retrosigmoid approach has not been reported. In our patient, it is possible that at the time of placement of the lazy-S-shaped incision the trigeminal nerve was stimulated in the retroauricular area overlying the dermatome supplied by the mandibular nerve root (V3). [Figure 2] illustrates a surface reconstructed image of the patient, showcasing the patient’s surgical scar obtained from post-operative magnetic resonance imaging superimposed on the innervation of the face and scalp. The image highlights the overlapping region between the surgical incision and the area supplied by the V3 root.
Zhang et al. have identified risk factors associated with the TCR which suggests that patients with pre-operative hypertension may not be as susceptible to developing TCR due to potentially higher sympathetic tone and lower vagal activity.[6] On the other hand, the use of calcium channel blockers (CCBs) has been shown to be associated with TCR, and in our case, the patient had recently been started on Cilnidipine. Compression of the brainstem by a CP angle lesion is another risk factor for the occurrence of TCR.[7]
Our case report makes a significant contribution to the existing literature on brain-heart interactions. First, it highlights that TCR can occur right from the skin incision even in the retrosigmoid lateral suboccipital approach to the CP angle where the majority of the incision is away from the trigeminal nerve distribution. Second, though hypertension is not directly associated with TCR occurrence, our patient with pre-operative hypertension experienced TCR. Thus, other factors such as the patient’s pre-operative medications (in this case CCB) and tumor compression of the brainstem may have contributed. When managing these cases under TCI without muscle relaxation, the presence of apnea should also be recognized as a potential finding alongside the expected hemodynamic changes. The BP of the patient decreased below the pre-operative level after tumor resection, suggesting a potential link between the involvement of the brainstem and the underlying cause of hypertension.
CONCLUSION
Brain-heart interactions in CP angle surgeries can be evident from an early stage including the pre-operative presence of raised BP. The occurrence of TCR at the skin incision should serve as an early warning sign, emphasizing the need for heightened vigilance during the subsequent steps of the surgical procedure.
Ethical approval
The Institutional Review Board approval is not required.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship
Nil.
References
- Trigeminal cardiac reflex: New thinking model about the definition based on a literature review. Medicine (Baltimore). 2015;94:e484.
- [CrossRef] [PubMed] [Google Scholar]
- Trigeminocardiac reflex during surgery in the cerebellopontine angle. J Neurosurg. 1999;90:215-20.
- [CrossRef] [PubMed] [Google Scholar]
- Management of the trigeminocardiac reflex: Facts and own experience. Neurol India. 2009;57:375-80.
- [CrossRef] [PubMed] [Google Scholar]
- Trigemino-cardiac reflex may be refractory to conventional management in adults. Acta Neurochir (Wien). 2008;150:509-10.
- [CrossRef] [PubMed] [Google Scholar]
- Severe bradycardia and hypotension after connecting negative pressure to the subgaleal drain during craniotomy closure. Br J Anaesth. 2006;96:608-10.
- [CrossRef] [PubMed] [Google Scholar]
- Risk factors associated with trigeminocardiac reflex in patients with trigeminal neuralgia during percutaneous balloon compression: A retrospective cohort study. Clin Neurol Neurosurg. 2023;231:107834.
- [CrossRef] [PubMed] [Google Scholar]
- The influence of trigeminocardiac reflex on postoperative cardiac adverse events in patients undergoing cerebellopontine angle tumor resections: A case-control study. World Neurosurg. 2023;172:e291-8.
- [CrossRef] [PubMed] [Google Scholar]







