Translate this page into:
Cerebellopontine angle ependymoma in a 2-year- old child
Address for correspondence: Dr. Pravin Salunke, Department of Neurosurgery, PGIMER, Chandigarh - 160 012, India. E-mail: drpravin_salunke@yahoo.co.uk
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The management of cerebellopontine angle (CPA) ependymoma in children below 3 years of age is challenging. In addition to the rare occurrence of disease, the difficulty also lies in achieving gross total resection and providing radiotherapy in this subset of patients. We describe a case of CPA ependymoma in a 2-year-old child. Gross total excision was achieved followed by administration of radiotherapy. In this article, a review of literature for this rare entity and the difficulties faced in the surgery and adjuvant treatment has been discussed.
Keywords
Cerebellopontine angle
children
ependymoma
Introduction
Childhood cerebellopontine angle (CPA) ependymoma in children below 3 years of age is of rare occurrence with only a few studies addressing these.[1] Gross total resection is difficult to achieve and radiotherapy is generally withheld in this group of patients in view of side effects.
Case Report
A 2-year-old female child presented with complaints of vomiting with dullness for 15 days with involvement of lower cranial nerves. Radiology showed right cystic solid multiloculated CPA lesion, isointense on T1, hyperintense on T2 and enhancing on contrast [Figures 1a–f]. The patient underwent retro-mastoid sub-occipital craniectomy and near total excision. The tumor was reddish-gray fleshy, with cystic areas. It was moderately vascular with no apparent calcification, extending both posterior and anterior to the 7-8th nerve complex and could be dissected from the brain stem medially except close to the ponto-medullary junction. Post-operatively, the patient gradually started improving and finally became conscious again. The histopathology revealed grade II ependymoma showing perivascular pseudorosettes with a small component of rare foci of few bizarre cells with hyperchromatic nuclei and the number of mitoses were < 5 per 10 high power fields. Few areas of necrosis without palisading were encountered. GFAP positivity was mainly seen in the perivascular regions. The Ki67/MIB index was not done.
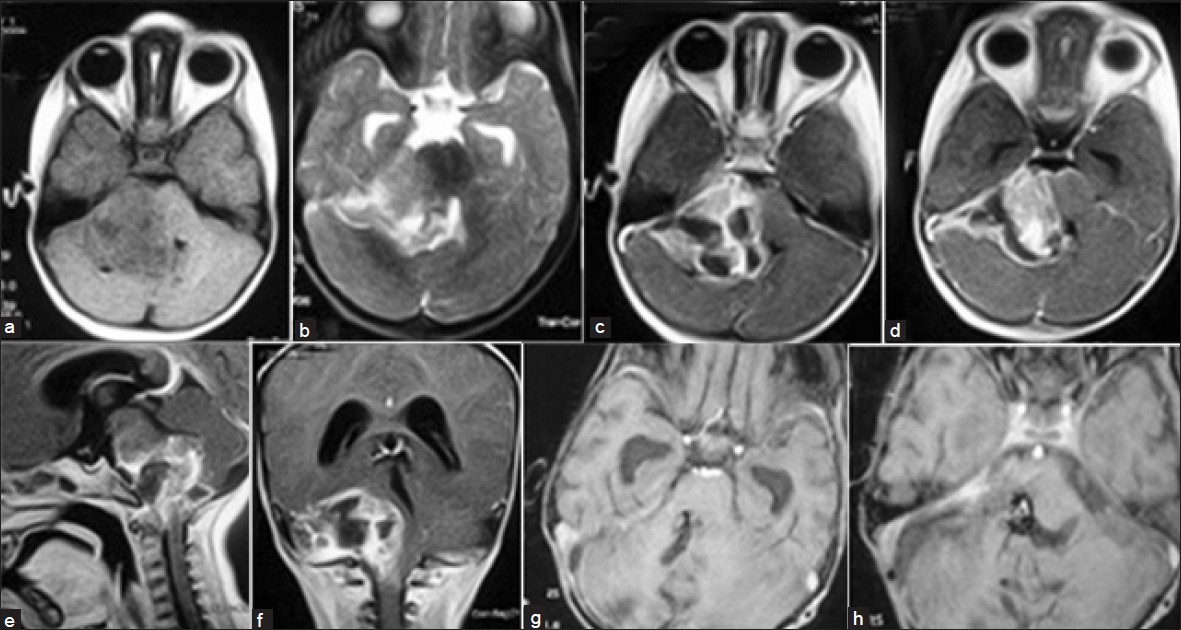
- (a) Axial T1-weighted image showing iso-hypointense lesion in the right cerebellopontine (CP) angle. (b) Axial T2-weighted image showing the extra-axial hyperintense lesion in the CP angle. (c and d) Contrast axial image showing the enhancing lesion with cystic areas compressing the brain stem and fourth ventricle. (e) Sagittal contrast magnetic resonance imaging (MRI) section showing the extent of lesion. (f) Coronal contrast MRI section showing the extent of lesion. (g) Axial contrast MRI showing gross total excision and opened fourth ventricle. (h) Axial contrast MRI showing gross total excision and the foramen of Luschka is clear
Post-op MRI (6 weeks later) was suggestive of complete excision of tumor [Figures 1g and h]. The child was subjected to focal radiotherapy but could not tolerate it beyond 7 days (10 grays in 5 fractions to the tumor bed). She had multiple episodes of vomiting. Child maintained her neurological status and gradually improved in general condition. Eight months later, she deteriorated due to parieto-occipital abscess at the shunt site and finally succumbed to ventriculitis.
Discussion
Childhood CPA ependymomas are uncommon variants of ependymomas of posterior fossa originating from ependymal cell rests which are present in the foramen of Luschka and appear to arise from the lateral surface of brain stem. They have a propensity to encase the lower cranial nerves and adjacent blood vessels namely Posterior Inferior Cerebellar artery. They grow laterally, further into CPA and medially into the IV ventricle. As they grow they displace the surrounding tissues bringing about rotation of the brain stem and unlike relatively commoner IV ventricular ependymomas, brain stem involvement does not occur at an early stage. This alters the anatomy and distorts the posterior fossa landmarks affecting the surgical approach and increases the operative difficulty.[1] Differential diagnosis of lesions at CPA includes certain common extra axial lesions like acoustic neuromas, meningiomas, vascular ectasia, and aneurysms. Less common lesions are epidermoid and other schwannomas as well as metastases, paragangliomas, and arachnoidal cysts. Intra-axial tumors in the area of the cerebellopontine angle include the medulloblastoma, astrocytoma, and the ependymoma.[2] The occurrence of a primary myxopapillary ependymoma and subependymoma at CPA has also been reported.[3–5] Mridha et al. have reported the occurrence of a metastatic lesion at CPA from lumbo-sacral myxopapillary ependymoma.[6]
Histologically, a variety of types, including cellular, clear cell, papillary, tanycytic, and myxopapillary have been described. Various forms of differentiation, including lipomatous, cartilaginous and osseous melanotic, neuronal and, most recently, sarcomatous have been described in ependymomas.[7]
They assume a large size before becoming symptomatic. Initial presentation is with features of raised ICP because of hydrocephalous. Involvement of cranial nerves and brain stem occurs at a later stage.[1]
CPA ependymomas on the plain CT scan appear as large extra axial masses in posterior fossa pushing the brain stem and fourth ventricle to the opposite side. Although no magnetic resonance imaging features are pathognomonic of ependymoma, these generally appear as lobulated tumors, which are hypointense on T1W images and hyperintense on T2W images, demonstrate irregular enhancement and are markedly heterogeneous due to calcification, hemorrhage, cystic components, or necrosis. T1 and T2 sequences are required to exactly delineate the encasement of vessels and to appreciate the rotation and positioning of brain stem for correct surgical planning.[18] Ependymomas in general have a propensity for neuraxis metastasis. However, no cases of neuraxis metastasis from CP angle ependymomas have been reported.[1]
Gross or near total excision (GTR) offers the patient maximal chances of prolonged overall survival (OS) as well as progression-free survival (PFS). Surgical resection in a young child remains a formidable task because of small size of posterior fossa, lower amount of blood, and significant difficulty in appreciating the local anatomy because of displacement of normal structures. Use of technological advances such as Cavitron Ultrasonic Aspirator (CUSA) and neurophysiologic monitoring would allow for total or near total resection of tumors. Post-operative MRI must be performed within 48 h of surgery to evaluate the extent of surgery. Second look surgery has been recommended to maximize the extent in the case of unexpected residual tumor or when the decision for a staged operation is made.[1] Adjuvant therapy of CPA ependymomas in age <3 years in the form of post-op radiotherapy although the standard line of treatment today based on results from the St. Jude RT-1 trial, is a new endeavor.[19] The group of patients with gross total or near total excision is recommended conformal radiotherapy. Focal irradiation for patients with localized ependymoma has also evolved. It administers higher doses of radiation (54-59.5Gy) to the tumor while sparing the surrounding tissues using a 10 mm clinical target volume and therefore beneficial in children below 3 years.[9] Proton radiotherapy although dosimetrically superior to photon radiotherapy is likely to become available after its clinical significance and population subset likely to benefit by its use is clearly defined. The role of neuraxis irradiation is limited to cases, which have proven metastasis at presentation.[9]
The best response rates to combination chemotherapy in primary ependymoma have been demonstrated in younger children who received pre-irradiation chemotherapy in an attempt to delay RT. In the POG 8633 infant study for patients less than 3 years of age, Duffner et al. reported a 48% response rate following two cycles of combination chemotherapy that consisted of vincristine, cyclophosphamide, cisplatin, and etoposide in 25 children with residual tumor after initial surgery, demonstrating that the use of post-operative chemotherapy may allow the delay of RT for a clinically relevant period of time in younger children with ependymoma.[10] Similarly, in a recent study by the French Society of Pediatric Oncology (SFOP), children with ependymoma under the age of 5 were treated with seven cycles of alternating courses of procarbazine and carboplatin, etoposide and cisplatin, vincristine and cyclophosphamide over 1.5 years.[11] Despite the lack of any partial (PR) or complete responses (CR) in patients with residual disease post-initial surgery, 23% of patients remained alive at 4 years without the use of RT, suggesting that there exists a small subset of patients for whom cure may be possible with surgery and post-operative chemotherapy alone.[12]
Presently, chemotherapy is reserved for those with subtotal resection (STR) before they are taken up for second look surgery.[13]
Extent of tumor resection remains the single most important prognostic factor for childhood CPA ependymoma. The 5 year probability of OS in patients with GTR is 67-80% and 5 year PFS as 51-75%.[1] As compared to this, 5 year PFS of patients with STR is 22-47%. This strongly signifies the role of additional resection for residual tumor, if any. Children under 3 years of age experience poor survival.[3] This has been explained previously by the fact that gross total resection (GTR) is accomplished less frequently in CPA surgery in young children. In addition, radiotherapy at age <3 years is generally withheld, delayed, or reduced in intensity, further reducing chances of long-term survival. Significance of histological grade continues to be debated and remains controversial.[1415]
Ependymomas in pediatric age are usually classified in WHO grade II (Low grade or differentiated ependymomas) or grade III (high grade or anaplastic ependymomas). Foci of hypercellularity, nuclear pleomorphism and anaplasia, necrosis, mitoses > 10 per 10 high power field and vascular proliferation qualify for grade III. The histological grade predicts the event free survival but not the overall survival.[15] Proliferating indices have a role on patients’ survival rates. Definition of cut-off between benign and aggressive forms also remains controversial, although the cutoff for Ki-67 has been fixed at 20.5%. These limits are used to define low (<20.5%) and high (>20.5%) Ki-67 indices and predict favorable (>5 years survival) or unfavorable (<5 years) patients outcome.[16]
Cytogenetic studies in one third of cases show deletions or rearrangements of 6q, 17, and 22 and gain of 1q chromosomes are detected, Gain of 1q25 indicates a poor prognosis.[14]
In conclusion, CPA ependymomas in children <3 years are rare and its successful management remains elusive. The mainstay of treatment in CP angle ependymoma in children <3 years is gross total resection followed by conformal radiotherapy which enables higher doses to be given with least side effects. Prophylactic radiotherapy to neuroaxis has role only to those who have proven metastasis. The role of chemotherapy either neo-adjuvant or following the surgery in these cases is still not defined.
Source of Support: Nil
Conflict of Interest: None declared.
References
- Advances in surgical techniques for resection of childhood cerebello-pontine angle ependymomas are key to survival. Childs Nerv Syst. 2009;25:1229-40.
- [Google Scholar]
- Differential diagnosis of space demands in the cerebellopontine angle. Radiologe. 2004;44:1113-36.
- [Google Scholar]
- Myxopapillary ependymoma of the cerebellopontine angle: Retrograde metastasis or primary tumour? Br J Neurosurg. 2011;25:122-3.
- [Google Scholar]
- Primary myxopapillary ependymoma of the cerebellopontine angle: Report of a case. Neurosurg Rev. 2009;32:241-4.
- [Google Scholar]
- Subependymoma of the cerebellopontine angle and prepontine cistern in a 15-year-old adolescent boy. AJNR Am J Neuroradiol. 2008;29:190-1.
- [Google Scholar]
- Myxopapillary ependymoma of lumbosacral region with metastasis to both cerebellopontine angles: Report of a rare case. Childs Nerv Syst. 2007;23:1209-13.
- [Google Scholar]
- Ependymal tumours with sarcomatous change (“Ependymosarcoma”): A clinicopathologic and molecular cytogenetic study. Am J Surg Pathol. 2008;32:699-709.
- [Google Scholar]
- Images in Neuro Oncology: Primary extraaxial cerebellopontine angle ependymoma. J Neurooncol. 2007;83:31-2.
- [Google Scholar]
- Preliminary results from a phase II trial of conformal radiation therapy and evaluation of radiation-related CNS effects for pediatric patients with localized ependymoma. J Clin Oncol. 2004;22:3156-62.
- [Google Scholar]
- Postoperative chemotherapy and delayed radiation in children less than three years of age with malignant brain tumors. N Engl J Med. 1993;328:1725-31.
- [Google Scholar]
- French Society of Pediatric Oncology.Postoperative chemotherapy without irradiation for ependymoma in children under 5 years of age: A multicenter trial of the French Society of Pediatric Oncology. J Clin Oncol. 2001;19:1288-96.
- [Google Scholar]
- New chemotherapy strategies and biological agents in the treatment of childhood ependymoma. Childs Nerv Syst. 2009;25:1275-82.
- [Google Scholar]
- Ependymoma: New therapeutic approaches including radiation and chemotherapy. J Neurooncol. 2005;75:287-99.
- [Google Scholar]
- Survival following treatment for intracranial ependymoma: A review. Childs Nerv Syst. 2009;25:1303-12.
- [Google Scholar]
- The prognostic value of histological grading of posterior fossa ependymomas in children: A Children's Oncology Group study and a review of prognostic factors. Mod Pathol. 2008;21:165-77.
- [Google Scholar]
- Ki-67 immunolabeling index is an accurate predictor of outcome in patients with intracranial ependymoma. Am J Surg Pathol. 2004;28:914-20.
- [Google Scholar]






