Translate this page into:
A Randomized Controlled Trial on the Study of Effectiveness and Safety of Hormonal (ACTH) Treatment Alone versus Hormonal (ACTH) with Levetiracetam for Epileptic Spasms
Vykuntaraju K. Gowda, DM Department of Pediatric Neurology, Indira Gandhi Institute of Child Health Near NIMHANS, Bengaluru, Karnataka 560029 India drknvraju08@gmail.com
This article was originally published by Thieme Medical and Scientific Publishers Pvt. Ltd. and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Abstract
Background West's syndrome (WS) is a triad of epileptic spasms (ESs), psychomotor delay, and hypsarrhythmia. The treatment of ESs is still controversial. Hence, we designed a randomized controlled trial (RCT) to compare the outcomes in children with WS treated with adrenocorticotropic hormone (ACTH) alone versus ACTH and levetiracetam (LEV).
Objectives To compare the treatment outcomes and side effects in children treated with ACTH alone versus ACTH and LEV.
Methods This prospective randomized controlled trial was conducted from December 2017 to May 2019 in tertiary care center, Bangaluru. Children from 2 months to 5 years of age, diagnosed with WS were included. Fifty children in each group were analyzed for efficacy and side effects.
Results There was no difference in the baseline characteristics in both groups. There was no difference in spasms response at the end of 2 weeks between the groups (88 vs. 82%) with p-value of 0.813. The relapse rates were less in ACTH and LEV group (20%) compared with ACTH alone (22%) but statistically not significant (p > 0.1). There was no difference observed in subsequent epilepsy rates (18%) in ACTH versus 19% in ACTH with LEV group (p > 0.1) and side effects. There was improvement in milestones 48% in ACTH with LEV group versus 37% in ACTH alone however statistically not significant (p > 0.1).
Conclusion There was no difference in children treated with ACTH alone versus ACTH and LEV in terms of control of spasms and subsequent epilepsy rates. The relapse rate is less, and developmental outcome is better in ACTH with LEV group but statistically not significant.
Keywords
epileptic spasms
levetiracetam
West's syndrome
ACTH
Introduction
Epileptic spasms (ESs) are an age-specific epileptic syndrome of infancy and early childhood. Children with ES typically exhibit ESs along with the electroencephalographic (EEG) pattern known as hypsarrhythmia. West in 1841 was the first to describe West's syndrome (WS).1 WS comprises a triad of spasms, psychomotor delay, and hypsarrhythmia. Incidence is 1 in 2,000 to 3,000 live births.2 3 Peak of onset is in the first year of life.
It is unclear as to what is the ideal treatment for ESs, but most studies suggest hormonal therapy, adrenocorticotropic hormone (ACTH) or glucocorticoids and vigabatrin.4 Role of other antiepileptics drugs (AEDs) in the treatment of ES is uncertain. Recently, levetiracetam (LEV) has been found to be a suitable drug for investigation in the treatment of ES as it is a broad-spectrum AED including ES; also, it has added benefit of favorable kinetics and negligible systemic toxicity.5 6 7 8 Early identification and aggressive treatment are important in this devastating disorder as it causes neuroregression without treatment.9 In the recent study by O'Callaghan et al showed that combination of hormonal treatment with vigabatrin versus hormonal treatment alone is more effective.9 Only few studies (Gümüş et al,8 five children and Mikati et al,5 seven children) studied usage of LEV in ES. The most common cause of ES in India is symptomatic etiology, requiring long-term medications, despite ESs are controlled with steroids. Many treating physicians start broad-spectrum AEDs before or along with hormonal treatment for ES as these children are prone for other seizures due to structural pathology. Lesser lead time is associated with better outcome, hence rapid diagnosis and effective treatment of ES are required. Since overall methodology of the studies till date has been poor, optimal drug in the treatment of ES is yet to be clear.10 Hence, the current randomized controlled trial (RCT) was designed to compare the efficacy and adverse events in children treated with ACTH alone versus ACTH and LEV.
Methods
The study was conducted in a tertiary-level health center, south India. The source of data included children diagnosed with WS attending pediatrics neurology inpatient and outpatient departments. Children between the age group of 2 months and 5 years, diagnosed with WS based on ES, psychomotor delay, and interictal EEG showing hypsarrhythmia or its variants were included. Children with WS secondary to tuberous sclerosis, biotinidase deficiency, and pyridoxine dependency were excluded; also children who were already treated previously and children with contraindication for steroids such as infections and hypertension were also excluded from the study groups. It was a prospective randomized interventional study conducted from December 2017 to May 2019.
Informed consent was taken from the parents of children. Details of history and physical examination were recorded in a predesigned, pre-structured, and pretested pro forma. Baseline developmental status was assessed by developmental assessment scale for Indian infants (DASII). Interictal EEG was done in all children in awake and sleep periods. Neuroimaging—computed tomography/magnetic resonance imaging was done in children wherever required to establish the etiology. Screening of inborn errors of metabolism—arterial blood gas analysis, lactate, ammonia, tandem mass spectrometry, gas chromatography mass spectrometry, cerebrospinal fluid glucose and lactate, and others were done whenever needed and feasible, also in all children with cryptogenic and idiopathic WS . Pyridoxine trial was given to all cryptogenic and idiopathic WS cases, and nonresponders were enrolled in the study.
Children, who were included in the study, were randomly assigned into two groups by computerized randomization. One group was allocated for intervention with hormone ACTH and another group was allocated for intervention with hormone ACTH and LEV. Initial screening tests were done to rule out infections. After initial assessment and investigations, randomization was done with computer-generated tables and children received either injection ACTH alone or injection ACTH with LEV. Patients were assessed on 14 days for response to the treatment in their respective treatment groups and classified into responders and nonresponders depending on cessation of spasms. All were followed up at regular interval—1 month, 3 months, and 6 months, and outcomes were measured in terms of response, relapses, if any, improvement in milestones, subsequent epilepsies. Side effects of drugs such as gastritis, weight gain, hypertension, hyperglycemia, somnolence, and agitation were noted.
In ACTH group, injection ACTH 100 units per body surface area intramuscular or subcutaneous (IM/SC) was given for 2 weeks and response was assessed at the end of 2 weeks, upon response, oral steroids were added for 2 weeks and tapered and stopped over a period of 4 weeks. If no response, other alternative treatment was initiated. In ACTH and LEV groups, injection ACTH 100 units per body surface area IM/SC was given for 2 weeks with oral LEV in two doses per day, 20 mg/kg/d for the first two doses and increased to 30 mg/kg/d after 24 hours, and if spasms continued after 72 hours, dosage was increased to 40 mg/kg/d. If child responded, oral steroids were added for 2 weeks and tapered and stopped over a period of 4 weeks. LEV was continued.
Primary clinical response was measured in terms of percentage of cessation of spasms at the end of 2 weeks as no response, partial response, and complete response if <25, 25– < 100, and 100% of cessation of spasms, respectively. All children during follow-up were monitored for relapse in both the intervention groups. Development delay was analyzed as a measure of the deviation from the chronological age and was measured at 1, 3, and 6 m postentry into the trial. Development was assessed by DASII scale. Patients were also monitored for side effects such as gastritis, weight gain, hypertension, hyperglycemia, somnolence, and agitation. Drugs were discontinued only if serious or life-threatening events occurred subsequent epilepsy rate: All children during follow-up were observed for development of other seizure types irrespective of their incidence before or at the time of enrollment into the study.
Various definitions11 were used in this study. The term “West syndrome” was used to describe the combination of ESs in single or clusters along with hypsarrhythmia on EEG. ESs were used to describe the clinical spasms that occur in clusters. The clinical spasms included brief synchronous movements of head, trunk, and limbs, that lasted longer than movements of myoclonus and shorter than movements associated with tonic seizures, with an average duration of 1 second. Hypsarrhythmia was used to describe the pattern on EEG characterized by random, high voltage spikes and slow waves that arise from many foci. Modified hypsarrhythmia was used to describe variants of hypsarrhythmia, which included burst suppression pattern, multifocal spikes, and synchronized bursts of generalized spikes with some preservation of background rhythm. Etiological classification was followed as per 2017 International League Against Epilepsy (ILAE) classifications.12 The term clinical relapse was defined as any episode of spasms that occurred in clusters; or two or more episodes of spasms that occurred singly and not in clusters. It was considered at any stage after primary clinical response was obtained. However, a single episode of single spasm was not considered a relapse.
Sample Size Calculation
There are no previous studies comparing ACTH versus ACTH with LEV. O'Callaghan et al9 reported cessation of spasms in combination and hormonal therapies was 89 and 69%, respectively. Hence to estimate and compare the prevalence of cessation of spasms between combination and hormonal therapies and to achieve a power of 80% for detecting a difference in proportions of 20% between the two groups (test—reference group) at a one-sided p-value of 5%, the required sample size is 50 in each group. The sample size was calculated using nMaster 2.0 software.
Statistical Analysis
Data were entered using Excel and analyzed using R software version 3.6.1. All categorical information was presented as frequency and percentages. All continuous measurements were summarized using mean (standard deviation) or median (interquartile range) based on the distribution. All categorical clinical parameters between ACTH and ACTH + LEV groups was analyzed using chi-square or Fisher's exact test based on their expected frequency. All continuous measurements were assessed using independent sample t-test or Mann–Whitney's U test based on the normal distribution assumption. For all comparison, p-value is considered significant at 5% level of significance.
Results
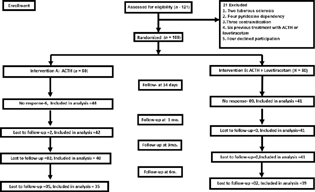
A total of 121 children were enrolled in the present study. Out of which 21 patients were excluded as per exclusion criteria. The remaining 100 children were randomly allocated into two interventional groups, who received either ACTH or ACTH and LEV and were assessed at 2 weeks for cessation of spasms and responders, that is, 44 in ACTH group and 41 in ACTH and LEV group were followed at regular intervals of 1 month, 3 months, and 6 months. Fig. 1 shows flow of patients in the study population.
-
Fig. 1 Flow chart of study population. ACTH, adrenocorticotropic hormone.
Fig. 1 Flow chart of study population. ACTH, adrenocorticotropic hormone.
Table 1 shows comparison of baseline characteristics in both the groups. The p-value was >0.05, and thus, no statistically significant difference was noted between the two groups. Of the 100 children included in the study, only 6 children (6%) had normal development prior to the onset of spasms. Regression achieved milestones in 8% children in each group.
|
Baseline characteristic |
ACTH (n = 50) |
ACTH + LEV (n = 50) |
p-Value |
||
|---|---|---|---|---|---|
|
Mean |
Percentage or IQR |
Mean |
Percentage or IQR |
||
|
Age of presentation (mo) |
11 |
7, 16 |
10 |
8, 13 |
0.79 |
|
Sex: Male:female |
39:11 |
78:22 |
34:16 |
68:32 |
0.260 |
|
Age of onset of ES (mo) |
5 |
3, 8 |
6 |
3, 9 |
0.521 |
|
Semiology |
|||||
|
Flexor:extensor:mixed |
42:6:2 |
84:12:4 |
46:3:1 |
92:6:2 |
0483 |
|
Clusters: Yes:no |
45:5 |
90:10 |
45:5 |
90:10 |
0.999 |
|
Relationship with the sleep–wake cycle |
|||||
|
Yes:no |
39:11 |
78:22 |
40:10 |
80:20 |
0.629 |
|
Spasm load |
7 |
4, 10 |
5 |
4, 8 |
0.134 |
|
Lead time to treatment (mo) |
3 |
2, 9 |
3 |
1, 7 |
0.435 |
|
Gestation: term:preterm |
44:6 |
88:12 |
45:5 |
90:10 |
0.749 |
|
Birth weight |
|||||
|
AGA:SGA:LGA |
34:16:0 |
68:32:0 |
34:14:2 |
68:28:4 |
0.548 |
|
Perinatal insult |
|||||
|
Hypoglycemia |
8 |
16 |
7 |
14 |
0.779 |
|
Birth asphyxia |
24 |
48 |
19 |
38 |
0.313 |
|
Development prior to the onset of spasms |
|||||
|
Delayed:normal |
48:2 |
96:4 |
46:4 |
92:8 |
0.679 |
|
Visual impairment: Yes:no |
25:25 |
50:50 |
17:33 |
34:66 |
0.105 |
|
Hearing impairment: Yes:no |
10:40 |
20:80 |
7:43 |
14:86 |
0.424 |
|
Neuroregression: Yes:no |
4:46 |
8:92 |
4:46 |
8:92 |
0.999 |
|
Microcephaly: Yes:no |
38:12 |
76:24 |
36:14 |
72:28 |
0.648 |
|
Number of children on antiepileptic drugs before initiation of intervention medications |
|||||
|
Yes:no |
23:27 |
46:54 |
24:26 |
48:52 |
0.841 |
Abbreviations: ACTH, adrenocorticotropic hormone; AGA, appropriate for gestational age; ES, epileptic spasm; IQR, interquartile range; LEV, levetiracetam; LGA, large for gestational age; SGA, small for gestational age.
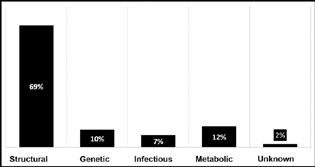
The most common cause of etiology was structural 69 (69%), followed by metabolic 12 (12%), genetic 10 (10%), and infectious 7 (7%). Only 2 (2%) cases were of unknown etiology where no underlying cause could be established even after a detailed work-up. Fig. 2 shows distribution of etiology in children according to the ILAE Commission for Classification and Terminology 2017. Of the 69 cases of structural etiology, hypoxic-ischemic encephalopathy (HIE) was the most common cause (41%), followed by hypoglycemia (9%), HIE and hypoglycemia (9%), and malformation of brain (7%). Metabolic etiology was seen in 12% of the cases, of which, four were Leigh's disease, four of them had organic acidemia, two had phenylketonuria, and one other had storage disorder. The EEG done at the time of admission revealed hypsarrhythmia in 9 (18%) and modified hypsarrhythmia in 41 (92%) children ACTH group. In ACTH and LEV groups, hypsarrhythmia was found in 10 (20%) and modified hypsarrhythmia in 40 (80%) cases.
-
Fig. 2 Distribution of etiology of epileptic spams according to the International League Against Epilepsy Commission for Classification and Terminology 2017.
Fig. 2 Distribution of etiology of epileptic spams according to the International League Against Epilepsy Commission for Classification and Terminology 2017.
Primary response was assessed at the end of 14 days of start of treatment. It was observed that 54% showed complete response in each group. A total of 31% patients showed partial response (17 patients in ACTH and 14 patients in ACTH + LEV group). Fifteen of 100 patients showed nil response (6 patients in ACTH and 9 patients in ACTH + LEV group), p-value = 0.641. Table 2 shows response to therapy, relapse and lost to follow-up, and developmental outcome. It was observed that there was no statistically significant difference between the two treatment arms. After excluding nonresponders, 44 in ACTH group and 41 in ACTH and LEV group were followed up. It was observed that there was no statistically significant difference between the two interventional groups with respect to relapse rates as seen by p-value > 0.1, developmental outcome (p > 0.1). Eight out of 44 (18.20%) in ACTH group and 8 out of 41 (19.50%) in ACTH and LEV group subsequently developed other forms of epilepsy during follow-up. Statistically no significant difference was noted in the two treatment groups (p = 0.902) (Table 3). There is no statistically significant difference between the two groups.
|
Response |
ACTH (n = 50) |
ACTH + LEV (n = 50) |
p-Value |
|---|---|---|---|
|
Spasms response on day 14 |
N = 50 |
N = 50 |
|
|
No response |
6 (12%) |
9 (18%) |
0.813 |
|
Partial |
17 (34%) |
14 (28%) |
|
|
Complete |
27 (54%) |
27 (54%) |
|
|
At 1 mo |
N = 42 |
N = 41 |
|
|
Relapse |
15 (35.70%) |
9 (21.90%) |
0.76 |
|
Lost to follow-up |
2 (4.80%) |
0 |
|
|
At 3 mo |
N = 40 |
N = 41 |
|
|
Relapse |
6 (15%) |
11 (26.80%) |
0.31 |
|
Lost to follow-up |
2 5% |
0 |
|
|
At 6 mo |
N = 35 |
N = 39 |
|
|
Relapse |
1 (2.90%) |
0 |
0.53 |
|
Lost to follow-up |
5 (14.30%) |
2 (5.10%) |
|
|
Assessment of milestones on follow-up |
|||
|
1 mo |
42 (%) |
41 (%) |
|
|
Gaining milestones |
18 (42.90) |
22 (53.70%) |
0.42 |
|
Lost to follow-up |
2 (4.8) |
0 |
|
|
3 mo |
N = 40 |
N = 41 |
|
|
Gaining milestones |
22 (55%) |
30 (73.20%) |
0.37 |
|
Lost to follow-up |
2 (5%) |
0 |
|
|
6 mo |
N = 35 |
N = 39 |
|
|
Gaining milestones |
13 (37.10%) |
19 (48.70%) |
0.28 |
|
Lost to follow-up |
5 (14.30%) |
2 (5.10%) |
|
Abbreviations: ACTH, adrenocorticotropic hormone, LEV, levetiracetam.
|
Subsequent other epilepsy |
ACTH (n = 44) |
ACTH + LEV (n = 41) |
p-Value |
|---|---|---|---|
|
Present |
8 (18.20%) |
8 (19.50%) |
0.902 |
|
Side effects |
ACTH N = 50, N (%) |
ACTH + LEV N = 50, N (%) |
|
|
Gastritis |
|||
|
14 d |
29 (58) |
36 (72) |
0.99 |
|
Last, follow-up |
11 (22) |
13 (26) |
|
|
Weight gain |
|||
|
14 d |
7 (14) |
4 (8) |
0.97 |
|
Last, follow-up |
11 (22) |
5 (10) |
|
|
Hyperglycemia |
0 |
0 |
0.00 |
|
Hypertension |
0 |
0 |
0.00 |
|
Somnolence |
|||
|
14 d |
0 |
11 (22) |
0.99 |
|
Last, follow-up |
0 |
16 (32) |
|
|
Agitation and behavioral problems |
|||
|
14 d |
0 |
6 (12) |
0.99 |
|
Last, follow-up |
0 |
9 (18) |
|
Abbreviations: ACTH, adrenocorticotropic hormone, LEV, levetiracetam.
Discussion
Patients with ES do not respond to treatment with conventional AEDs.13 Guidelines and reviews recommend treatment with hormones such as ACTH and vigabatrin.10 14 15 16 However, spasms persist in 33 to 56% of patients despite the use of these effective treatments.17 18 LEV is commonly prescribed broad-spectrum AEDs before initiation of hormonal treatment. Moreover, treatments used at present vary in most institutions because of factors such as cost, adverse effect profiles, or availability of recommended drugs, indicating the need for well-designed clinical trials. To date, there have been few well-designed RCTs that have considered the treatment of ESs.10
The average age of presentation of children with ES in the current study was 10.5 months. This was like other Indian studies.19 20 21 However, some Western studies22 23 24 showed slightly early presentation, probably because of increased awareness to seek treatment at an early stage. Table 4 shows comparison of age of presentation, age of onset, and lead time (in month) in various studies for ES. The average age of onset of spasms in the present study was found to be 5.5 months. This was in concordance with other studies, with maximum children within classical age of onset, that is, between 3 and 9 months. The earliest age of onset noted in our study was 1 month and one child presented with onset as late as 25 months. The lead time to treatment is 3 months. The lead time was longer when compared with Western studies23 25 but was in concordance with other Indian studies.19 20 This delay was probably owing to delay in seeking medical attention by parents due to ignorance, lack of awareness, late referral to concerned specialist due to the subtleness of its nature, as well as use of inappropriate AEDs such as phenytoin, and carbamazepine.
|
Study |
Intervention |
Age of presentation (mo) (mean) |
Age of onset of spasms (mo) (mean) |
Lead time (mo) (mean) |
|---|---|---|---|---|
|
Current study |
ACTH ACTH + LEV |
11 10 |
5 6 |
3 3 |
|
Gowda et al19 |
Prednisolone ACTH |
13.9 9.4 |
6 8.6 (7.3) |
5.23 3.35 |
|
Baram22 |
ACTH and prednisone |
6 |
– |
– |
|
Yanagaki et al23 |
ACTH (high dose) ACTH (low dose) |
7 10.2 |
5 5.7 (5.3) |
1.7 4 |
|
Ibrahim et al24 |
ACTH and vigabatrin |
6.5 ± 2.3 |
5 ± 1.4 (5) |
1.4 ± 1.4 |
|
Lagae et al25 |
ACTH and vigabatrin |
12.2 ± 8.9 |
7 ± 3 |
− |
|
Kaushik et al20 |
– |
13.1 ± 7.3 |
5.3 ± 4.6 |
7.9 ± 7.4 |
|
Lagae et al25 |
– |
– |
– |
6 d |
Abbreviations: ACTH, adrenocorticotropic hormone; LEV, levetiracetam.
The current study showed a male preponderance, with boys accounting for 73%. This was in concordance with other Indian studies 62,19 81,20 and 54%.21 This again could be because of gender-biased referral and treatment-seeking behavior of parents. This gender bias is well rooted in the Indian society where male children are brought to medical attention more frequently and earlier as compared with their female counterparts.
We observed that flexor type of ES (88%) is commonest type like other studies 7321 and 65%.20 In the current study, most of the children (97%) were developmentally delayed which was supported by other studies 93,20 94,19 and 73.24 This brings us to the conclusion about the significance of early recognition and treatment of ES to prevent further delay in attaining age-appropriate milestones.
Since the etiological classification in our study revealed predominantly a structural etiology (69%), an associated feature of this being microcephaly was seen in 74% compared with 65%.26 Among the etiological subtypes, symptomatic variety was the most common in the present study, accounting for 88% of cases, followed by idiopathic cases (10%) and cryptogenic (2%). The percentage of symptomatic cases was like Indian studies19 24 but slightly higher when compared with developed countries studies.11 22 24 25 This probably reflects the poor maternal and neonatal health services in developing countries. Most of the studies failed to distinguish between cryptogenic and idiopathic types.
At the end of 2 weeks of treatment, no statistically significant difference was found between the two groups in controlling spasms p-value (0.813). Table 5 shows comparison of cessation of ES in various studies. In the current study, the response to ACTH was 88% as was seen with other studies such as 86.6,22 82,11 and 76%.27 There are no studies done with the combination of ACTH and LEV, for comparison, although a few studies done were with LEV as a monotherapy for the treatment of ES. Sharma and Vishwanthan21 tried LEV in a total of nine children with ES and found that five (55.5%) of them showed partial response to the drug, while four (44.5%) of them did not show any cessation of spasms. No child showed a full response in this study. Gümüş et al8 studied response to LEV in five children diagnosed with cryptogenic WS. Out of the five children, two (40%) of them showed complete response, two responded partially, and one did not respond to the treatment. Mikati et al5 enrolled seven children with ES and observed that two (28.6%) showed complete response and rest five (71.4%) improved partially.
|
Study |
Prednisolone |
ACTH |
ACTH + levetiracetam |
|||
|---|---|---|---|---|---|---|
|
Present study |
– |
– |
44/50 |
88% |
41/50 |
82% |
|
Gowda et al19 |
6/15 |
40% |
11/18 |
61.11% |
– |
– |
|
Hrachovy et al27 |
7/24 |
29.16% |
9/24 |
37.5% |
– |
– |
|
Azam et al11 |
51/72 |
71% |
27/33 |
82% |
– |
– |
|
Lux et al30 |
21/30 |
70% |
19/25 |
76% |
– |
– |
|
Baram22 |
4/14 |
28.6% |
13/15 |
86.6% |
– |
– |
|
Wanigasinghe et al28 |
32/48 |
66.7% |
20/49 |
40.8% |
– |
– |
|
Levetiracetam |
||||||
|
Sharma and Vishwanthan21 |
– |
– |
– |
– |
5/9 |
55.55% |
|
Gümüş et al8 |
– |
− |
– |
– |
4/5 |
80% |
|
Mikati et al5 |
– |
– |
– |
– |
7/7 |
100% |
Abbreviation: ACTH, adrenocorticotropic hormone.
Overall relapse rate was low in ACTH and LEV group (17.93%) in comparison to ACTH group (21.66%), though not statistically significant. The relapse rates in ACTH group were similar like other studies.19 23 The relapse rate in ACTH and LEV group in our study is low (17.93%) compared with ACTH and valproate group (31.80%) in a study done by Sharma and Vishwanthan.21 Overall, it was observed that children in ACTH and LEV group had better improvement in milestones compared with ACTH group, though it was proved statistically insignificant. In a study by Gowda et al,19 36.36% children showed improvement by gaining new milestones, though not achieved completely normal development for that age. At the end of 6 months, out of 85 children with ES, 18.9% children developed epilepsy during their subsequent follow-up visits. This was low in comparison to other studies: 79.20,11 48.30,22 39.40,19 and 58%.21 Low rates of subsequent epilepsies in our study were probably because 50% of the children in our study were on other AEDs prior to the start of treatment.
Though gastritis was observed in a greater number of children in both the groups, it was not severe enough to discontinue the therapy. Wanigasinghe et al28 observed increased appetite (43.2%), weight gain (31.20%), frequent crying spells (25%), cushingoid features (20.50%), drowsiness (15.90%), dyspepsia (4.5%), and vomiting (2.3%) as common side effects in their study. The most reported side effects associated with the use of LEV are somnolence and fatigue.29 Less frequent side effects consist of agitation, aggression, anxiety, or depression.13 Side effects associated with LEV use in our study were somnolence in and agitation. No children in either of the group required discontinuation of therapy owing to the side effects.
Limitations
Outcome was measured as a clinical response of cessation of spasm. Electroclinical response with resolution of hypsarrhythmia, which is a better indicator of outcome, was not considered. It would have been desirable to do a 24-hour EEG recording to document cessation of spasms and resolution of hypsarrhythmia. However, it was not done due to the lack of resources. Clinical resolution of spasms may be an acceptable approach in a resource-poor setting. Observer and analyzer bias could not be eliminated due to absence of blinding. The study lacked exceptionally long-term follow-up to determine final developmental outcome.
Conclusion
No difference was found in control of spasms, relapse rate, developmental outcome, and subsequent epilepsy rates between the two treatment groups; however, relapse rate is less, and developmental outcome is better in ACTH with LEV group but statistically not significant. Delay in identification and diagnosis has been found to be significant, and emphasizes the need to increase awareness of this entity among general practitioners. More than 80% of children responded to hormonal treatment. HIE was the leading cause of ES, which is preventable one, hence emphasizing the need of improvement in our maternal and child health services.
Conflict of Interest
None declared.
Funding None.
References
- The descriptive epidemiology of infantile spasms among Atlanta children. Epilepsia. 1999;40(6):748-751.
- [Google Scholar]
- Epidemiologic features of infantile spasms in Iceland. Epilepsia. 1994;35(4):802-805.
- [Google Scholar]
- Infantile spasms–evidence based medical management. Indian J Pediatr. 2014;81(10):1052-1055.
- [Google Scholar]
- Effect of levetiracetam in refractory childhood epilepsy syndromes. Eur J Paediatr Neurol. 2003;7(3):123-128.
- [Google Scholar]
- Levetiracetam in the treatment of infantile spasms. Eur J Paediatr Neurol. 2005;9(1):19-22.
- [Google Scholar]
- Levetiracetam monotherapy in newly diagnosed cryptogenic West syndrome. Pediatr Neurol. 2007;37(5):350-353.
- [Google Scholar]
- Safety and effectiveness of hormonal treatment versus hormonal treatment with vigabatrin for infantile spasms (ICISS): a randomised, multicentre, open-label trial. Lancet Neurol. 2017;16(1):33-42.
- [Google Scholar]
- Treatment of infantile spasms (review) The Cochrane Collaboration. ; 6: [Internet] [cited October 4, 2015]. Accessed April 15, 2020 at: http://www.thecochranelibrary.com
- [Google Scholar]
- Use of ACTH and prednisolone in infantile spasms: experience from a developing country. Seizure. 2005;14(8):552-556.
- [Google Scholar]
- ILAE classification of the epilepsies: position paper of the ILAE Commission for Classification and Terminology. Epilepsia. 2017;58(4):512-521.
- [Google Scholar]
- Recent advances in the pharmacotherapy of infantile spasms. CNS Drugs. 2014;28(4):279-290.
- [Google Scholar]
- Summary of recommendations for the management of infantile seizures: Task Force Report for the ILAE Commission of Pediatrics. Epilepsia. 2015;56(8):1185-1197.
- [Google Scholar]
- Evidence-based guideline update: medical treatment of infantile spasms. Report of the Guideline Development Subcommittee of the American Academy of Neurology and the Practice Committee of the child Neurology Society. Neurology. 2012;78(24):1974-1980.
- [Google Scholar]
- The epilepsies: the diagnosis and management of the epilepsies in adults and children in primary and secondary care. ; Clinical guideline 137
- [Google Scholar]
- Vigabatrin versus ACTH as first-line treatment for infantile spasms: a randomized, prospective study. Epilepsia. 1997;38(12):1270-1274.
- [Google Scholar]
- A risk-benefit assessment of treatments for infantile spasms. Drug Saf. 2001;24(11):813-828.
- [Google Scholar]
- Corticotrophin-ACTH in comparison to prednisolone in West syndrome - a randomized study. Indian J Pediatr. 2019;86(2):165-170.
- [Google Scholar]
- Clinical spectrum and treatment outcome of West Syndrome in children from Northern India. Seizure. 2013;22(8):617-621.
- [Google Scholar]
- Treatment of infantile spasms: the ideal and the mundane. Epilepsia. 2003;44(8):993-994.
- [Google Scholar]
- A comparative study of high-dose and low-dose ACTH therapy for West syndrome. Brain Dev. 1999;21(7):461-467.
- [Google Scholar]
- Clinical profile and treatment of infantile spasms using vigabatrin and ACTH–a developing country perspective. BMC Pediatr. 2010;10:1.
- [Google Scholar]
- Treatment and long term outcome in West syndrome: the clinical reality. A multicentre follow up study. Seizure. 2010;19(3):159-164.
- [Google Scholar]
- Ontogeny of corticotropin releasing hormone gene expression in rat hypothalamus–comparison with somatostatin. Int J Dev Neurosci. 1991;9(5):473-478.
- [Google Scholar]
- High-dose, long-duration versus low-dose, short-duration corticotropin therapy for infantile spasms. J Pediatr. 1994;124:803-806. (5 Pt 1):
- [Google Scholar]
- Randomized, single-blind, parallel clinical trial on efficacy of oral prednisolone versus intramuscular corticotrophin on immediate and continued spasm control in West syndrome. Pediatr Neurol. 2015;53(3):193-199.
- [Google Scholar]
- United Kingdom Infantile Spasms Study. The United Kingdom Infantile Spasms Study (UKISS) comparing hormone treatment with vigabatrin on developmental and epilepsy outcomes to age 14 months: a multicenter randomized trial. Lancet Neurol. 2005;4(11):712-717.
- [Google Scholar]







