Translate this page into:
Bladder dysfunction in Indian patients with neurological disorders – An observational study
*Corresponding author: Samhita Panda, Department of Neurology, All India Institute of Medical Sciences, Jodhpur, Rajasthan, India. samhitapanda@yahoo.com
-
Received: ,
Accepted: ,
How to cite this article: Agrawal N, Panda S, Bhaskar S, Yadav T, Tiwari S, Bhirud DP. Bladder dysfunction in Indian patients with neurological disorders – An observational study. J Neurosci Rural Pract. doi: 10.25259/JNRP_365_2024
Abstract
Objectives
There is a paucity of literature regarding bladder symptoms in neurological disorders, especially from India. While bladder symptoms are inquired into during clinical interviews, significant importance and detailed assessment are rarely done. This study was planned to assess the spectrum of neurogenic bladder dysfunction encountered in a hospital-based setting in India.
Materials and Methods
This cross-sectional observational study was performed over 18 months. Patients more than 18 years old with bladder dysfunction owing to neurological disorders without obstructive pathology were included. Details regarding clinical symptoms, bladder involvement, and examination findings were gathered, and a neurogenic bladder symptom score (NBSS) questionnaire was administered.
Results
A total of 72 patients (mean age 48.8 ± 17.46 years) were enrolled with neurological disorders such as extra-medullary lesions of the spinal cord, demyelinating disorders, parkinsonian disorders, and dementia among others. Clinical localization of bladder to frontal level was found in 11.1%, suprapontine (34.7%), suprasacral (37.5%), sacral/infra-sacral (9.7%), and upper motor neuron bladder with shock (7.0%). NBSS provided a fairly good idea of severity in respective domains, i.e., incontinence, storage and/or voiding, and complications associated with bladder symptoms. However, this was not directed toward the initial assessment of suspected neurogenic bladder and diagnostic level of localization of bladder symptoms. In addition, some questions of NBSS are not tailored to the Indian population and habits.
Conclusion
This study reflects the spectrum of neurogenic bladder dysfunction in Indian patients. With limitations in the applicability of available bladder questionnaires to the Indian population, a tool aimed at evolving a diagnostic formulation of the level of bladder involvement will be beneficial, especially with the advent of tele-neurology.
Keywords
Incontinence
Localization
Neurogenic bladder
Storage
Voiding
INTRODUCTION
Lower urinary tract symptoms (LUTSs) comprise a combination of storage, voiding, and postmicturition symptoms. The physiological process of micturition includes cyclical storage and voiding of urine which is coordinated by the interplay of activity between the two important parts of the lower urinary tract through an intricate neural regulatory system located in the brain, spinal cord, and peripheral ganglia: (a) The bladder acting as the reservoir for storage and (b) the outflow tract including the neck of the bladder, urethra and it’s sphincter, and prostate gland in men.[1]
Neurological disorders leading to neurogenic LUTS include traumatic spinal cord injury (SCI), demyelinating disorders, parkinsonian disorders, stroke, cauda equina syndrome, and diabetes mellitus.[2] An uninhibited bladder with lessened awareness of bladder fullness and low-capacity bladder is caused by suprapontine lesions, i.e., above the pontine micturition Centre (PMC) such as in stroke, dementia, or brain tumor. Upper motor neuron (UMN) bladder or automatic bladder results from damage to structures between PMC and sacral level of the spinal cord, i.e., infra-pontine and suprasacral location such as in traumatic SCI or myelitis. The disruption of alternate contraction of detrusor and sphincter muscles leading to detrusor-sphincter dyssynergia (DSD) classically represents UMN neurogenic bladder dysfunction producing high bladder pressures causing vesicoureteral reflux (VUR). In addition, neurogenic detrusor overactivity (NDO) causes decreased bladder capacity and detrusor muscle hypertrophy also enabling VUR and incontinence. Lower motor neuron (LMN) or autonomous bladder is caused by damage to either the sacral spinal cord or peripheral motor and sensory innervation of the bladder, suggesting sacral or infrasacral location.[2,3]
In view of the fact that such a vast range of neurological conditions has an influence on the role of the lower urinary tract, the resulting urinary dysfunction is variable. Few patients having neurogenic lower urinary tract dysfunction (LUTD) suffer from symptoms associated with impaired storage of the urine, including the increase in the frequency of micturition, urgency, and incontinence of urine and few may have symptoms related to voiding such as hesitancy, slowing of the urinary stream, straining while urination and retention of the urine. The type, site, and extent of the neurological lesion and its progression determine the severity and nature of the neurogenic bladder.[3,4] Symptoms related to the urinary tract impact the quality of life (QOL) of a person significantly. For instance, they lead to compromise in activities of daily living and cause humiliation, in turn leading to social isolation and an increase in the social and healthcare-related burden of the society.[5]
Literature is scant regarding bladder symptoms in neurological disorders from India. While bladder symptoms may be inquired into during the clinical interview, significant importance and detailed assessment is rarely done. Most of the questionnaires related to the neurogenic bladder are either for QOL assessment or severity assessment. The neurogenic bladder symptom score (NBSS), which has good validity and reliability, has also not been widely studied in all subsets of neurological disorders in India.[6] Therefore, this study was planned to assess the spectrum of neurogenic bladder dysfunction encountered in a tertiary care hospital-based setting and the use of NBSS for assessment of bladder dysfunction in neurological disorders.
MATERIALS AND METHODS
This cross-sectional, observational single-center study was carried out in the Department of Neurology. The study was undertaken after due ethical clearance by the Institutional Ethics Committee (AIIMS/IEC/2021/3683). Patients attending outpatient and in-patient services from July 2021 to December 2022 satisfying the inclusion criteria and exclusion criteria given below were enrolled after informed written consent.
Inclusion criteria
Patients with the age of 18 or more
Patients having neurological disorders associated with bladder dysfunction
Patients who were able to give the relevant history of bladder dysfunction and undergo the relevant tests
Patients willing to provide written informed consent.
Exclusion criteria
Patients in altered sensorium mandating catheterization and/or in intensive care unit setting
Patients with disorders in comprehension preventing appropriate history from being elicited
Patients with historical complaints of urinary tract infection or urological issues such as benign prostatic hyperplasia, bladder/urethral injury, cystitis, kidney/bladder stone, or any other obstructive cause
Patients with previous urologic surgery.
Demographic details of patients such as name, age, sex, residence, education, and occupational status were obtained. Detailed clinical history with general physical and neurological evaluation comprising mini-mental status examination, motor and sensory system, cranial nerves, and cerebellum examination was done to assess the duration of neurological symptoms with the type of neurological disorder and the clinical level of involvement.
Symptoms related to bladder dysfunction were recorded. The type and level of bladder lesions were classified into frontal, suprapontine, suprasacral infrapontine, and sacral or infra-sacral lesions based on features in the existing literature.[3,7,8] In addition, the NBSS questionnaire was filled out for each patient (or by the caregiver if the patient was not able to) and total scores for each domain were calculated. The NBSS is a discriminative tool developed for patients with SCI, multiple sclerosis (MS), and spina bifida.[6] It is a 24-element questionnaire that has been already validated and measures bladder symptoms across three different domains: Incontinence (0–29), storage and voiding (0–22), and consequences (0–23); and a single general urinary QOL question scored from 0 for pleased to 4 for unhappy. For each domain, a larger score implies a worse symptom severity or QOL.[6]
Urine routine microscopy and urine culture sensitivity were done in all patients to look for urinary tract infections. Post-void residual volume (PVRV) assessment by ultrasonography was performed in some patients. Radiological imaging like computed tomography/magnetic resonance imaging of the brain/spine was done as indicated for the patients. Urodynamic studies (UDS) were done in a few patients.
Statistical analysis
Collected data were analyzed using the Statistical Package for the Social Sciences (SPSS) 22.00 for Windows; SPSS Inc., Chicago, USA software. Continuous variables were represented by means and standard deviations along with 95% confidence intervals of the means were calculated. For each point of evaluation, data were statistically analyzed by applying a one-way analysis of variance test. The difference between the two groups was detected using a t-test and Chi-square test with the level of significance set at P < 0.05.
RESULTS
A total of 72 patients were enrolled over 18 months [Figure 1]. The mean age of subjects was 48.8 ± 17.46 years (70.8% male and 29.2% female). The predominant age group was 18–30 years (23.6%) while 18.0% were between 31 and 40 years and 16.7% of 51–60 years. Most of the subjects (76.4%) presented with symptoms of >3 months duration while 16.7% were acute (≤30 days) and 6.9% subacute.
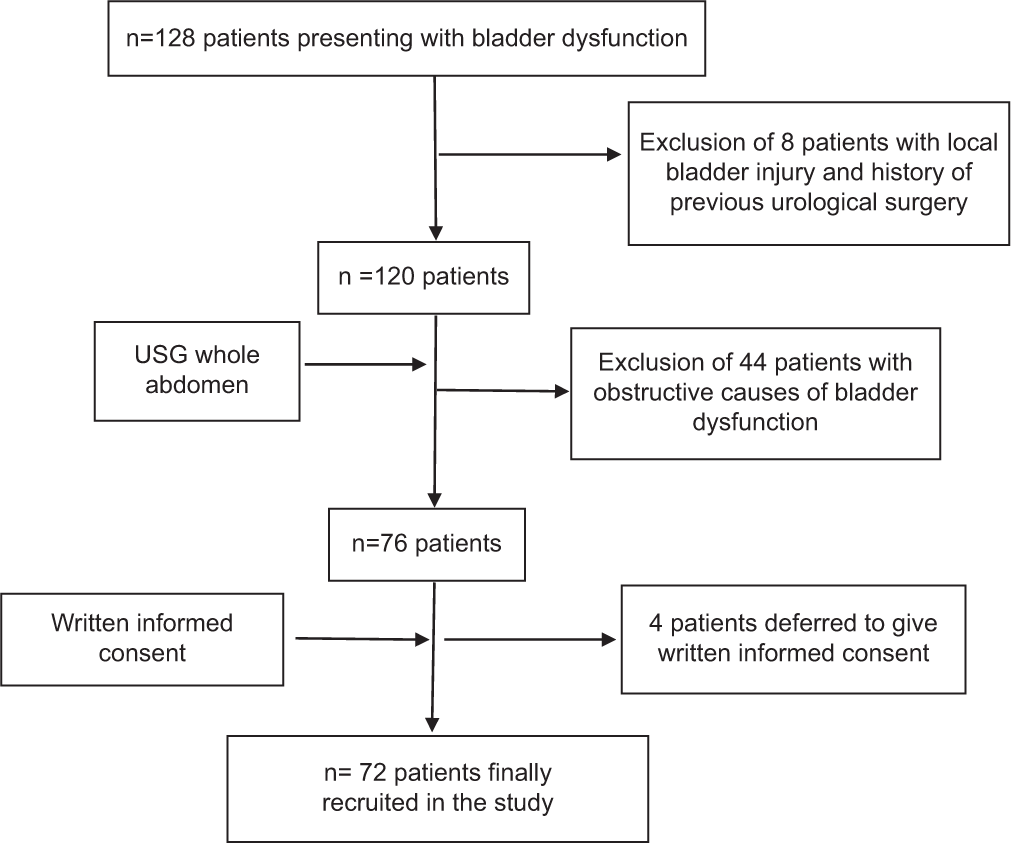
- Flowchart of recruitment of subjects into the study.
Neurological disorders observed included extra-medullary pathologies of the spinal cord (31.9%), demyelinating diseases (22.2%), Parkinson’s and related disorders (16.7%), dementia (12.5%), and others (16.7%). Based on a detailed clinical examination, the clinical level of the neurological lesion was deduced. The most common clinical level of the lesion was found at the cortical with the subcortical region (27.8%) followed by the lower cervical cord region (C5-C8) (16.7%) and subcortical alone (15.3%).
The presenting symptoms related to bladder dysfunction were grouped into symptom categories such as urinary urgency, frequency, urge incontinence, hesitancy, and others which were present in 70.8%, 65.2%, 62.5%, and 40.3%, respectively [Table 1]. Based on these, the study subjects were further categorized into the following groups of bladder dysfunction – storage, voiding, incontinence, functional incontinence, and decreased/absent bladder sensation. These were reported among 77.8%, 37.5%, 76.4%, 11.1%, and 15.3%, respectively [Figure 2]. Urinary incontinence that is not caused by LUTD characterizes functional incontinence that happens in cases of cognitive impairment which makes it difficult for the patient to reach the toilet in time, or the absence of an adequate number of attempts, as occurs in vascular dementia.
| Bladder symptoms | n | % |
|---|---|---|
| Urinary urgency | 51 | 70.8 |
| Urinary frequency | 47 | 65.2 |
| Urge incontinence | 45 | 62.5 |
| Urinary hesitancy | 29 | 40.3 |
| Loss of insight into bladder symptoms | 8 | 11.1 |
| Voiding at inappropriate places | 8 | 11.1 |
| Loss of bladder sensation | 11 | 15.3 |
| Feeling of incomplete evacuation | 26 | 36.1 |
| Dribbling of urine | 3 | 4.1 |
n: Number of subjects
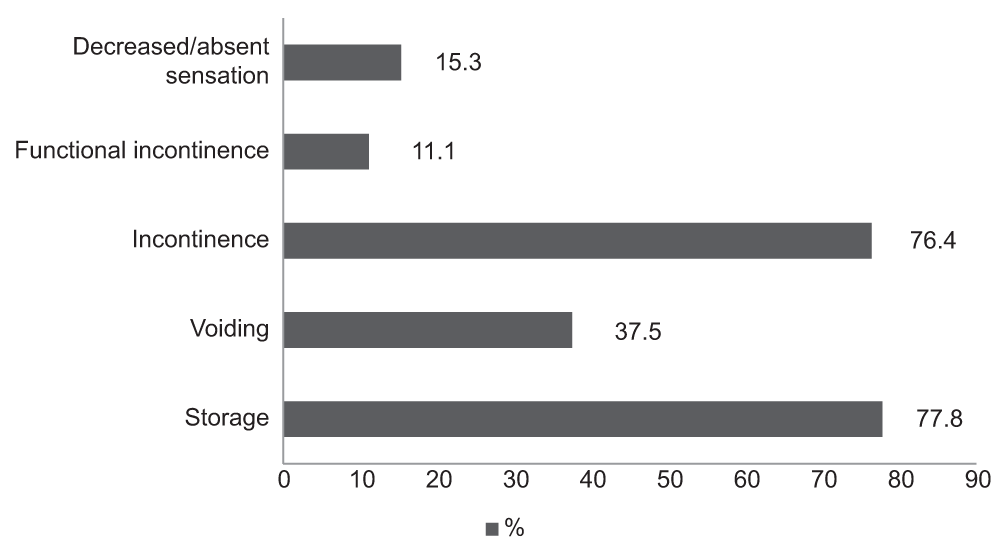
- Distribution of various categories of bladder dysfunction among the subjects.
Subsequently, the patients were finally classified into the following neurogenic bladder levels, i.e., frontal (11.1%), suprapontine (34.7%), suprasacral infrapontine (37.5%), sacral/infrasacral/peripheral arc (9.7%), and UMN bladder with shock (7.0%). Among these groups, suprasacral infrapontine was the most common urological localization for bladder symptoms followed by suprapontine.
On observing the types of catheterization methods in different neurogenic bladders, we found that all patients of frontal and suprapontine and most suprasacral infrapontine bladder types voided spontaneously. Indwelling catheter was mostly used in patients with suprasacral infrapontine, sacral/infrasacral bladder, and UMN bladder with shock and clean intermittent catheterization was used only in a few patients of suprasacral infrapontine and sacral/infrasacral bladder type [Supplementary Table 1].
Ultrasound of kidneys, ureters, and bladder showed changes of cystitis and hydronephrosis as a complication of bladder dysfunction in 4.2% each. Although patients did not complain of it, incidental urinary tract infection was present in 47.2%. Significant PVRV (>50 mL) was found in 29.2% of 34 subjects (due to lack of follow-up and absent bladder sensations in some patients) and mean PVRV was found to be 98.53 ± 156.95. Only 11 patients could undergo UDS (because of poor follow-up, recurrent urinary tract infection, and absent bladder sensation in some). NDO with DSD was found in 5 patients, NDO without DSD in 2 and underactive bladder in 4 patients.
Looking at neuro-radiological concordance with neurogenic bladder categories, all patients with frontal bladder type and most patients (21/25) with suprapontine bladder had imaging characteristics of cortical/subcortical involvement. Similarly, of 27 patients with suprasacral infrapontine bladder involvement, 21 had spinal cord lesions (cervical to thoracolumbar). Likewise, among 7 patients of LMN bladder (sacral/infrasacral), 6 had lesions along cauda equina/lumbosacral roots radiologically. On the basis of clinical, laboratory, and radiological evaluation, the final diagnosis arrived at in our study patients is given in Table 2.
| Final diagnosis | n | % |
|---|---|---|
| Vascular dementia | 7 | 9.7 |
| FTD | 2 | 2.8 |
| Huntington disease | 1 | 1.4 |
| Multiple sclerosis | 4 | 5.6 |
| Neuromyelitis optica spectrum disorder | 8 | 11.1 |
| Other demyelinating disorders | 4 | 5.6 |
| Spinocerebellar ataxia | 3 | 4.1 |
| Parkinson’s disease | 6 | 8.3 |
| Atypical Parkinson | 6 | 8.3 |
| Communicating hydrocephalus | 1 | 1.4 |
| Metabolic encephalopathy | 2 | 2.8 |
| Compressive myelopathy due to PIVD | 7 | 9.7 |
| Traumatic spinal cord injury | 6 | 8.3 |
| Cauda conus disorder | 4 | 5.6 |
| TB arachnoiditis | 2 | 2.8 |
| Anterior spinal artery thrombosis | 1 | 1.4 |
| Other non-compressive myelopathy (paraneoplastic/autoimmune/metabolic) | 3 | 4.1 |
| Mitochondrial ataxia | 1 | 1.4 |
| Leukodystrophy | 1 | 1.4 |
| Compressive myelopathy due to metastasis | 2 | 2.8 |
| Compressive myelopathy due to lumbar canal stenosis | 1 | 1.4 |
| Total | 72 | 100 |
n: Number of subjects, FTD: Frontotemporal dementia, PIVD: Prolapsed intervertebral disc, TB: Tuberculous.
The responses to the NBSS questionnaire are given in Figure 3a.[5] The NBSS aided in the categorization into only 3 domains, namely incontinence, storage and voiding, and consequences. NBSS helped in the assessment of the severity of neurogenic bladder symptoms. The mean values in the 3 major domains were 11.58 ± 9.92 (incontinence), 11.17 ± 5.29 (storage and voiding), and 3.85 ± 2.71 (consequences). The scores of incontinence and storage/voiding symptoms were mostly moderate to severe while consequences domain scores were milder.
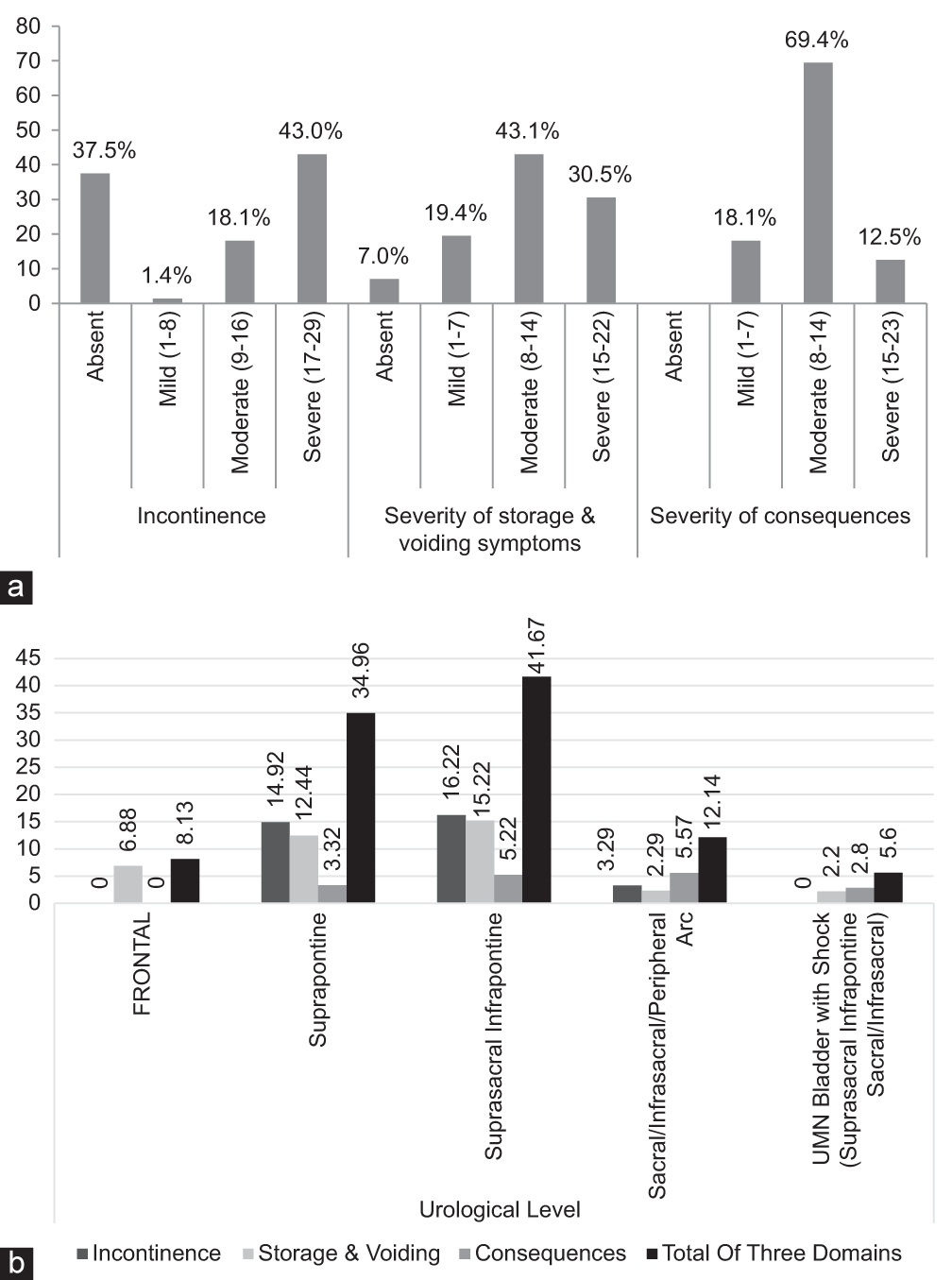
- (a) Severity of involvement in the domains of neurogenic bladder symptom score (NBSS) in the subjects (b) Distribution of the domains of NBBS questionnaire among the different levels of bladder involvement.
Comparison of urological localization of lesion with the NBSS domains revealed that suprasacral infrapontine and suprapontine bladder types were more significantly associated with incontinence and storage and voiding domains while consequences were with sacral/infrasacral and suprasacral infrapontine bladder types [Figure 3b]. However, the less severe affection of sacral/infrasacral and UMN bladder with shock types may be due to improper responses from patients because of decreased bladder sensations and indwelling catheterization. This may reflect some inherent fallacies of NBSS in appropriately assessing the severity of every bladder type. Indian subjects have restricted use of diapers due to unavailability, cost, convenience, and cultural habits, so some of the questions in NBSS could not be directly assessed, and a surrogate marker had to be used. In addition, the NBSS could not help in the evaluation of the level/type of bladder involvement. With respect to the effect on QOL, the NBSS questionnaire had a single question for its assessment that was graded on a Likert scale from 0 to 4 with 0 being pleased and 4 being unhappy. The mean value for the entire study group was 3.08 ± 0.81 with 12.5%, 56.9%, and 27.8% scoring 2, 3, and 4 on QOL, respectively, while only 2.8% were happy.
DISCUSSION
This study demonstrated the spectrum of bladder dysfunction in various neurological disorders in subjects from western Rajasthan. The symptoms of the neurogenic bladder are diverse and need to be probed in detail during the clinical interview to understand the site of involvement along the neuraxis as well as the nature, severity, and complications thereof so as to accord the most comprehensive treatment. Issues related to micturition are often discussed in hushed tones, especially in the social milieu existing in India. Hence, access to information related to this aspect of neurological disorders is largely pushed to the background by patients as well as physicians.
The gender bias with predominantly males (70.8%) presenting with neurogenic bladder symptoms may likewise represent hesitancy to discuss this openly compared to Western data (57.4% female).[9] Our study from the western part of India showed a younger age of presentation (48.89 ± 17.46 years) compared to Manack et al. (62.5 ± 19.6 years).[9] The distribution of neurological disorders observed was similar to other studies on the neurogenic bladder with the predominance of extramedullary spinal cord lesions including prolapsed intervertebral disc (9.7%) and traumatic SCI (8.3%) followed by demyelinating diseases (22.2%) and parkinsonian disorders (16.7% - Parkinson’s disease (PD) [8.3%] and atypical Parkinson’s disease [8.3%]). Manack et al. observed MS (20.1%) and SCI (9.0%) being the most frequent causes of neurogenic bladder while Ruffion et al. found that the prevalence of neurogenic LUTS was most commonly seen in SCI (52.3%) followed by MS (50.9%) and PD (33.1%).[9,10]
The clinical localization of the lesion according to the detailed history and neurological examination causing neurogenic bladder was most commonly localized to the dorso-lumbar spinal cord (23%), cervical spinal cord (22.2%), and subcortex (15.2%). Only 8.3% had involvement of cauda equina and lumbosacral roots. This corroborates with the involvement of dorso-lumbar segments of the spinal cord (sympathetic function) and/or subcortical PMC (Barrington nucleus) being crucial for bladder control. Similarly, in 56 patients of NTM with neurogenic bladder, the upper level of lesion in 71% of patients was above D10.[11] A meta-analysis of 1250 patients with traumatic SCI showed that 49.4% had involvement of the cervical cord and 47.8% of the dorsolumbar cord.[12]
In our study, most patients had UMN bladder symptoms such as urinary urgency, frequency, and urge incontinence.[7] This included 77.8% with symptoms related to storage such as increased frequency, urgency, and nocturia; 76.4% with incontinence; and 37.5% with obstructive urinary symptoms such as hesitancy, straining to void, and incomplete evacuation. LMN bladder symptoms such as decreased bladder sensation, overflow incontinence, and urinary retention were noted in only 15.3%. These findings also correspond with clinical and radiological levels of lesions, which are mostly UMN, i.e., cortical, subcortical, cervical, and dorsolumbar cords.
The NBSS is a patient-reported outcome measure that is valid and reliable.[6] In 2018, Welk et al. validated the use of NBSS in SCI patients for the assessment of neurogenic bladder symptoms and their QOL.[13] In 2015, Fragalà et al. used NBSS to demonstrate its correlation with UDS in patients of MS.[14] In 2019, Cintra et al. checked for the reliability and validity of NBSS in Brazilian Portuguese patients of MS and SCI with neurogenic bladder dysfunction.[5] However, it was certainly not used to evaluate the type and level of neurogenic bladder dysfunction. In our study, each domain of the NBSS was beneficial in portraying the range of bladder dysfunction and related complications.[15]
Comparison of the urological level of localization with the three domains of NBSS revealed that incontinence and storage and voiding domains’ scores were most severe in suprasacral infrapontine lesions (16.22 ± 8.7 and 15.22 ± 2.5, respectively) followed by lesions at suprapontine level (14.92 ± 7.8 and 12.4 ± 2.5, respectively), being statistically significant (P < 0.01). This implied that these bladder types are most severely affected according to NBSS. Of note, that for other types of neurogenic bladder including sacral/infrasacral and UMN bladder with shock, the mean scores for incontinence were 3.2 ± 8.6 and 0.00, respectively, and storage and voiding were 2.29 ± 2.7 and 2.2 ± 2.1, respectively. This would intuitively imply that these bladder types are less severely affected. However, this type of result could be fallacious due to the improper responses from the patients due to decreased bladder sensations or being catheterized early. Thus, this may highlight the fact that the NBSS cannot appropriately assess the severity of every type of neurogenic bladder.
At the same time, it is essential to acknowledge the constraints of the NBSS observed in this study. Assessment of the amount of urinary leakage during daytime in NBSS was on the basis of the number of pads used by the patients. However, in our subset of Indian patients, only a few of them used pads and most of them responded to the question on the basis of the amount of soaking of clothes or bed wetting. Thus, NBSS is not directly applicable to Indian subjects. Furthermore, the appropriateness of one bladder management technique over another could not be determined on the basis of NBSS because a patient with significant incontinence who is voiding spontaneously had a worse score in comparison to a similar patient with an indwelling catheter who therefore had a better score as also observed in previous study.[6]
In addition, the NBSS cannot help in the evaluation of the level/type of bladder involvement. Of note, NBSS was never used for evaluation of neurogenic bladder in the Indian population although several other questionnaires were used for assessment of bladder dysfunction in this population. International Consultation on Incontinence Questionnaire-Female LUTSs was used to study the risk factors and the impact of urinary incontinence on QOL in women by Desai and Sabnis.[16] Another study by Khadke and Dhupkar to determine the percentage of overactive bladder and their health-related QOL in adult females with stress/urge/mixed urinary incontinence based on the results of the questionnaire for female urinary incontinence diagnosis was done using an overactive bladder questionnaire.[17] However, none of these studies were directed to localize the lesion related to bladder dysfunction.
CONCLUSION
Analysis of bladder dysfunction in neurological disorders in the Indian context can be challenging. Detailed bladder history and examination may help understand bladder-related symptoms with fairly good consistency with radiological evaluation. While questionnaires like NBSS help evaluate the severity and complications associated with bladder dysfunction, there are limitations in Indian subjects, and need for a more comprehensive and diagnostically targeted questionnaire is needed to aid in clinics and teleneurology.
Ethical approval
The research/study was approved by the Institutional Review Board at Institutional Ethics Committee AIIMS Jodhpur, number (AIIMS/IEC/2021/3683), dated September 11, 2021.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Use of artificial intelligence (AI)-assisted technology for manuscript preparation
The authors confirm that there was no use of artificial intelligence (AI)-assisted technology for assisting in the writing or editing of the manuscript and no images were manipulated using AI.
Financial support and sponsorship: Nil.
References
- Neural control of the lower urinary tract. Compr Physiol. 2015;5:327-96.
- [CrossRef] [PubMed] [Google Scholar]
- Lower urinary tract dysfunction in common neurological diseases. Turk J Urol. 2020;46:S70-8.
- [CrossRef] [PubMed] [Google Scholar]
- Neurogenic lower urinary tract dysfunction. Handb Clin Neurol. 2013;110:209-20.
- [CrossRef] [PubMed] [Google Scholar]
- Cross-cultural adaptation and validation of the neurogenic bladder symptom score questionnaire for Brazilian Portuguese. Int Braz J Urol. 2019;45:605-14.
- [CrossRef] [PubMed] [Google Scholar]
- The validity and reliability of the neurogenic bladder symptom score. J Urol. 2014;192:452-7.
- [CrossRef] [PubMed] [Google Scholar]
- Lower urinary tract dysfunction in the neurological patient: Clinical assessment and management. Lancet Neurol. 2015;14:720-32.
- [CrossRef] [PubMed] [Google Scholar]
- Epidemiology and healthcare utilization of neurogenic bladder patients in a US claims database. Neurourol Urodyn. 2011;30:395-401.
- [CrossRef] [PubMed] [Google Scholar]
- Systematic review of the epidemiology of urinary incontinence and detrusor overactivity among patients with neurogenic overactive bladder. Neuroepidemiology. 2013;41:146-55.
- [CrossRef] [PubMed] [Google Scholar]
- Urodynamic profile of patients with neurogenic bladder following non-traumatic myelopathies. Ann Indian Acad Neurol. 2013;16:42-6.
- [CrossRef] [PubMed] [Google Scholar]
- Prediction of bladder outcomes after traumatic spinal cord injury: A longitudinal cohort study. PLoS Med. 2016;13:e1002041.
- [CrossRef] [PubMed] [Google Scholar]
- The Neurogenic Bladder Symptom Score (NBSS): A secondary assessment of its validity, reliability among people with a spinal cord injury. Spinal Cord. 2018;56:259-64.
- [CrossRef] [PubMed] [Google Scholar]
- Association between the neurogenic bladder symptom score and urodynamic examination in multiple sclerosis patients with lower urinary tract dysfunction. Int Neurourol J. 2015;19:272-7.
- [CrossRef] [PubMed] [Google Scholar]
- Clinical study of urinary incontinence in women at a tertiary care centre. Med Innov. 2021;10:10-4.
- [Google Scholar]
- Prevalence of overactive bladder syndrome among adult females having stress urinary incontinence and urge urinary incontinence using Overactive Bladder Syndrome questionnaire. Int J Health Sci Res. 2023;13:55-68.
- [CrossRef] [Google Scholar]






