Translate this page into:
Idiopathic hypertrophic pachymeningitis presenting with a superficial soft tissue mass
Address for correspondence: Dr. Inuka Kishara Gooneratne, 10/1 Borella Cross Road, Colombo 8, Sri Lanka. E-mail: kishig@gmail.com
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Idiopathic hypertrophic pachymeningitis (IHP) is a chronic progressive diffuse inflammatory fibrosis of the dura-mater, leading to its diffuse enlargement. The following describes a case of IHP presenting with a superficial soft tissue mass. A 40-year-old female came to hospital with a subcutaneous lump over the left face and frontal headache for 6 months. An excision biopsy revealed chronic inflammation. Magnetic resonance imaging (MRI) of the brain showed left mastoiditis and early dural inflammation of the left temporal region. A few months later, she developed diplopia, complex partial seizures, and retrobulbar neuritis of the left optic nerve. Repeat MRI brain demonstrated meningeal thickening on both sides of the tentorium cerebelli extending to the left tempero-parietal meninges. The meningeal biopsy revealed markedly thickened fibro-connective dural tissue with infiltration of chronic inflammatory cells. There was no evidence of bacterial, fungal, tuberculous or neoplastic infiltration. IHP was diagnosed and steroid therapy initiated. Within weeks, she showed marked clinical improvement. IHP is a diagnosis of exclusion. The absence of underlying infective, neoplastic, or systemic autoimmune disease favors IHP. The above patient had headache, neuro-ophthalmic signs, seizures, which are features of IHP. However, superficial soft tissue involvement is rare.
Keywords
Idiopathic hypertrophic pachymeningitis
tumefactive fibro-inflammatory lesion
tolosa-hunt syndrome
Introduction
Idiopathic hypertrophic pachymeningitis (IHP) is a chronic progressive diffuse inflammatory fibrosis of the dura-mater, leading to its diffuse enlargement. This rare disorder is usually found intracranially though spinal forms have also been described. The following describes a case of IHP presenting with a superficial soft tissue mass.
Case Report
A 40-year-old woman with an unremarkable past medical history presents with a swelling over the left face just above the tempero-mandibular joint and progressive bi- frontal headache for 6 months. The subcutaneous lump was 2.5 cm in diameter, firm, and non-tender. She had no other organ involvement. Her physical examination including neurology was normal except for the above-mentioned lump. Histology after the complete excision biopsy of the lesion revealed chronic inflammatory changes and fibrotic changes. MRI of the brain showed left mastoiditis and early dural inflammation of the left temporal region. The subsequent CT-guided dural biopsy also showed evidence of chronic inflammation with minimal fibrosis. Treatment was not initiated as the patient was asymptomatic and there was no recurrence of the lump after excision.
A few months later, she developed diplopia, complex partial seizures, and retrobulbar neuritis of the left optic nerve. Repeat magnetic resonance imaging (MRI) brain at this juncture demonstrated progressive disease, with meningeal involvement on both sides of the cerebral hemispheres [Figure 1a] and prominent extension to involve the left tempero-parietal meninges [Figure 1b]. Craniotomy of the corresponding region showed marked meningeal thickening spanning approximately 6 mm, with adherent thick exudates. The histology revealed markedly thickened dural fibro-connective tissue with focal infiltration of chronic inflammatory cells [Figure 2]. Fibrotic changes were more evident compared to the previous histology signifying progression. There was no evidence of bacterial, fungal, tuberculous, or neoplastic infiltration on histology. The erythrocyte sedimentation rate was elevated (95 mm/h, normal 1-20 mm/h) but other blood investigations, bone marrow trephine, chest X ray, ultra sound abdomen, serum calcium, CSF studies, skin pathergy, and mantoux were all normal. Rheumatoid factor, ANA, cANCA, and pANCA were within normal limits.
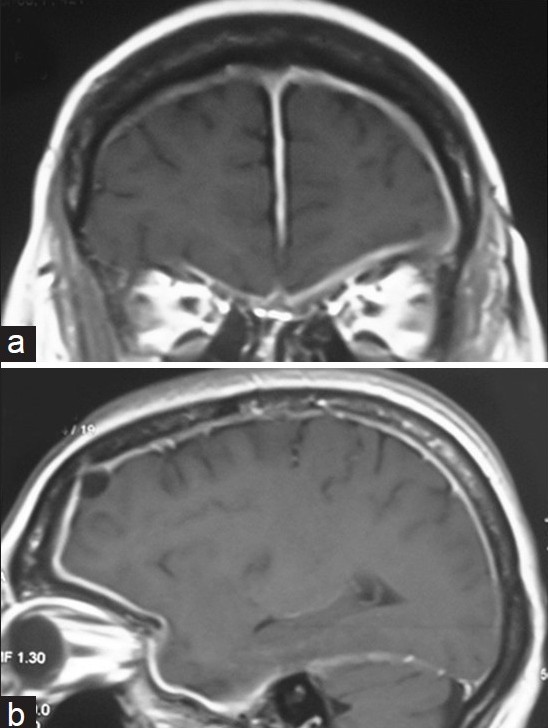
- (a) Enhanced T1-weighted MR (coronal) image shows thickened meninges extending over both cerebral hemispheres on either side of the falx cerebri with prominent extension to the left tempero-parietal region. (b) Enhanced T1-weighted MR (sagittal) image shows thickened meninges involving the left tempero-parietal region
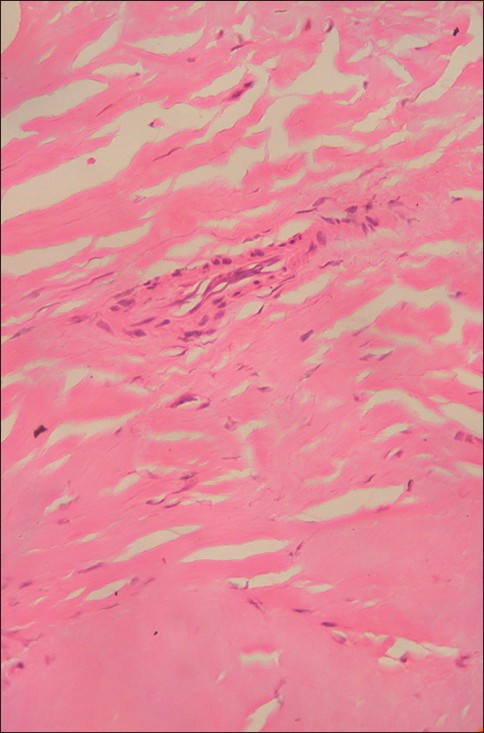
- A histopathologic examination reveals dense fibrosis with an acute and chronic inflammatory cell infiltration (arrow; H and E, ×400)
With the absence of demonstrable underlying infective, neoplastic, or systemic autoimmune disease, IHP was diagnosed and steroid therapy initiated. Within a few weeks, she showed marked clinical improvement. Currently, she is on a tapering dose of steroid therapy and is in remission.
Discussion
IHP is a diagnosis of exclusion.[1] The absence of demonstrable underlying infective, neoplastic, or systemic autoimmune disease favors IHP.[2] Common causes of hypertrophic pachymeningitis like sarcoidosis, tuberculosis, syphilis, fungal infections, lymphoma, Wegener's arteritis, rheumatoid arthritis, and other systemic autoimmune disorders need to be excluded.[3]
Cranial IHP can present with symptoms and signs of raised intracranial pressure, cranial nerve palsies, cerebellar ataxia, seizures, and neuro-ophthalmic symptoms such as visual field loss, complete blindness, and optic neuropathy.[3] Venous sinus thrombosis,[4] obstructive hydrocephalus,[4] and cerebral edema[3] are further documented complications of IHP. IHP also presents with chronic headache resembling migraine. Headache which is attributed to focal dural inflammation is a universal symptom, and at times may be the only symptom for many years. Intraparenchymal involvement in IHP is rare. The above-mentioned patient presented with headache, neuro-ophthalmic signs, and seizures.
MRI brain is useful in evaluating IHP. The thickened dura appears isointense to hypointense on both T1- and T2-weighted images and usually shows a uniform dense enhancement of the thickened membranes [Figures 1a and b]. The thickening is better appreciated on coronal and sagittal images. A thin rim of hyper-intensity may be present around the hypo-intense dura. Linear and nodular patterns of enhancement have been described with the former showing better therapeutic response, possibly due to lesser fibrosis and greater vascularity.[5]
Histopathological findings in IHP consist of thick fibrous dura often associated with chronic inflammatory cell infiltration consisting of lymphocytes, plasma cells, and histiocytes[6] [Figure 2]. No granuloma, microorganism, or evidence of vasculitis were detected in this case to suggest a recognized etiology. Histology of the soft tissue mass was identical suggesting the same disease process.
Superficial soft tissue involvement is an unusual presentation of IHP. A similar case by Holodny et al. reported a lesion that originated in the left side of the neck which progressed over 2 years to involve the meninges, the cavernous sinuses, the right temporal lobe, and the right orbit.[7] This lesion was described as a tumefactive fibro-inflammatory lesion (TFIL). Imaging studies of the lesion revealed a diffusely infiltrating mass arising from the left masticator space and extending in to the deeper structures of the neck. There was infiltration through the foramen ovale into the middle cranial fossa. Tissue from all these areas was histologically uniform and revealed a dense sclerosing, fibrous proliferative process, accompanied by a lymphoplasmacytic inflammatory infiltrate. Although similar in presentation, our patient did not demonstrate direct intracranial extension of the subcutaneous lesion. The authors go on to speculate on the relationship of TFILs to other inflammatory disorders of the head and neck, such as Tolosa-Hunt syndrome and IHP. The clinical, histological, and radiological similarities of these disorders to the case reported by Holodny as well as in the present case raise the possibility that these chronic inflammatory processes are spectra or variants of a single entity as opposed to discrete disorders.[7]
Due to the small number of cases of IHP identified early and the rarity of IHP, the natural history and prognosis of this disease entity are poorly documented. Spontaneous resolution has been reported.[89] The optimal management of IHP is unknown. The current standard of treatment is intended to curb the inflammatory response. Corticosteroid therapy is often effective in ameliorating the symptoms and signs, and in arresting the progression of the disease.[10] The patient in this case experienced significant improvement of symptoms after initiating steroids. Surgical excision is an option for patients with mass effect due to thickening of skull base dura mater who show no response to medical treatment.[11]
Hypertrophic pachymeningitis is a rare fibrosing inflammatory process involving the dura-mater and is a diagnosis of exclusion. The present case is unusual, with IHP manifesting as a soft tissue mass associated with meningeal involvement. This may represent a spectrum of a single chronic fibro-inflammatory process involving both subcutaneous and meningeal tissues.
Source of Support: Nil
Conflict of Interest: None declared.
References
- Idiopathic hypertrophic cranial pachymeningi tis associated with Sweet's syndrome. Eur J Radiol. 2002;44:139-42.
- [Google Scholar]
- Idiopathic hypertrophic spinal pachymeningitis: Report of two cases with typical mr imaging findings. AJNR Am J Neuroradiol. 2007;28:590-2.
- [Google Scholar]
- Hypertrophic Pachymeningitis: Varied Manifestations of a Single Disease Entity. Neurol India. 2002;50:45-52.
- [Google Scholar]
- Extensive anterior cranial fossa idiopathic hypertrophic pachymeningitis: A case report and review of the literature. J Med Assoc Thai. 2005;88:1934-40.
- [Google Scholar]
- Tumefactive Fibroinflammatory Lesion of the Neck with Progressive Invasion of the Meninges, Skull Base, Orbit, and Brain. AJNR Am J Neuroradiol. 2001;22:876-9.
- [Google Scholar]
- Spontaneous resolution of hypertrophic cranial pachymeningitis. Neurosurg Rev. 1995;18:201-4.
- [Google Scholar]
- Idiopathic tumefactive hypertrophic pachymeningitis. radiographics. 2005;25:1075-80.
- [Google Scholar]
- Hypertrophic pachymeningitis and ophthalmological disturbances: Description of two case reports. Arch Soc Esp Oftalmol. 2008;83:497-500.
- [Google Scholar]
- Idiopathic hypertrophic cranial pachymeningitis.Report of three cases. J Neurosurg. 1993;79:270-6.
- [Google Scholar]






