Translate this page into:
Glycopyrrolate-induced polyuria: An unexpected side effect
*Corresponding author: Pankaj Kumar Kannauje, Department of General Medicine, All India Institute of Medical Sciences, Raipur, Chhattisgarh, India. drpankajkannauje@aiimsraipur.edu.in
-
Received: ,
Accepted: ,
How to cite this article: Behera SK, Agarwal A, Singh A, Pandit VR, Kannauje PK. Glycopyrrolate-induced polyuria: An unexpected side effect. J Neurosci Rural Pract 2023;14:525-7.
Abstract
Polyuria is urine output exceeding 3 L/d in adults, primarily due to solute and water diuresis. In a hospital setting, mannitol and diuretics commonly lead to polyuria. We have found an interesting association of polyuria with glycopyrrolate; to the best of our knowledge, no case is reported in the literature. Here, we are describing a case of Guillain–Barre Syndrome, which developed polyuria during the hospital stay, which was secondary to glycopyrrolate.
Keywords
Polyuria
Glycopyrrolate
Guillain–barre syndrome
INTRODUCTION
Guillain–Barre syndrome (GBS) is a fulminant polyradiculopathy of acute onset manifesting as a rapidly progressing areflexic motor paralysis with dysautonomia, which may be associated with sensory disturbances.[1]
Glycopyrrolate is used to reduce airway or gastric secretions and abolish cardiac inhibitory reflexes during intubation. Glycopyrrolate is a peripherally acting anti-muscarinic agent which lacks blood–brain barrier permeability. It decreases the magnitude and acidity of gastric secretions and curbs excessive upper respiratory tract secretions. The common adverse effects are an extension of its pharmacological effects, which include xerostomia, urinary hesitancy and retention, blurred vision, photophobia, increased intraocular tension, and palpitation.[2] To the author’s knowledge, there is no documented case of glycopyrrolate-induced polyuria to date. We report a case of polyuria as a potential side effect of glycopyrrolate administration.
CASE REPORT
A 33-year-old female presented with a history of bilateral upper limb weakness, which progressed to involve bilateral lower limbs over 2–3 days. The patient had an episode of fever and loose stools lasting 2 days, 1 week before the symptom onset. Initially, the patient was conscious and oriented with intact higher mental functions. Furthermore, the power in different muscle groups was reduced, that is, 1/5 in the bilateral shoulder joint, 2/5 in the bilateral elbow, 2/5 in the small bilateral muscles of the hand, and 1/5 in the bilateral hip and knee joint. Bilateral knee, ankle, biceps, triceps, and supinator did not show deep tendon reflexes, while plantar reflex was also absent bilaterally. No sensory deficit was noted. Neither the bladder and bowel nor the cranial nerve were involved. The single breath count was six, and a provisional diagnosis of acute inflammatory demyelinating polyneuropathy was considered. Arterial blood gas analysis showed acidemia with respiratory acidosis with 7.253 and partial pressure of carbon dioxide 55.8 mm Hg, while bicarbonate and lactate were within normal limits. The patient was intubated, considering respiratory involvement. Other routine laboratory values, including liver and renal function tests, were unremarkable [Table 1]. High-sensitivity C-reactive protein was 64.14 mg/L. Cerebrospinal fluid analysis showed glucose 4.0 mmol/L, protein 201.16 mg/L, and a total cell count of zero, indicating albumin-cytological dissociation [Table 1].
| Test | Result | Reference Range |
|---|---|---|
| Analysis (ABG) | ||
| pH | 7.255 | 7.35–7.45 |
| pCO2 | 55.8 | 35–45 mm Hg |
| HCO3 | 24 | 22–26 mm Hg |
| Lactate | 0.57 | 0.5-2.2 mmol/L |
| Hematology | ||
| Hb | 11.1 | 12–15 g/dL |
| TLC | 11.08 | 4–10 × 109/L |
| Platelets | 159 | 150–410 × 109/L |
| Serum creatinine | 70.72 | 50–110 µmol/L |
| Serum potassium | 3.7 | 3.5–5 mmol/L |
| CRP | 64.14 | <10 mg/L |
| HbA1c | 5.2 | <6 mmoL/moL |
| AST | 24 | 12–38 U/L |
| ALT | 16 | 7–41 U/L |
| CSF | ||
| Glucose | 4.0 | 2.8–4.4 mmol/L |
| Protein | 201.16 | 20–40 mg/L |
| TLC | 00 | Nil |
ABG: Arterial blood gas, pCO2: Partial pressure of carbon dioxide, HCO3: Bicarbonate, Hb: Hemoglobin, TLC: Total leukocyte count, CRP: C-reactive protein, HbA1c: Glycated hemoglobin, AST: Alanine aspartate, ALT: Alanine transaminase, CSF: Cerebrospinal fluid analysis
The nerve conduction velocity study showed reduced amplitude of muscle action potential, absent F wave, and typical latency with severe axonal loss indicating acute motor axonal neuropathy variant. The diagnosis of GBS was confirmed. The patient was administered intravenous immunoglobulin (IVIG) at a dose of 0.4 g/kg/day for 05 days. After IVIG treatment, improvement in the weakness of both upper and lower limbs was observed, mechanical ventilation was gradually tapered, and the patient was extubated. On day 3 of extubation, the patient complained of continuous oral secretions. An opinion from the department of otorhinolaryngology was sought, which did not suggest any significant finding. Injection glycopyrrolate was added at a dose of 0.1 mg 8 hourly. The patient started having significantly increased urine output the next day. The day before adding glycopyrrolate, 24-h urine output was 2300 mL. After adding glycopyrrolate, urine output increased to 5600, 8300, and 6400 mL/day in subsequent days. No adverse hemodynamic consequences were noted. Urine osmolality came out as low as 64/kg of water. The patient’s daily fluid intake, serum glucose, or calcium remained the same throughout this time. Glycopyrrolate was discontinued because of polyuria. After discontinuation, 24-h urine volume was reduced to 1200 mL, and urine osmolality increased to 661 mOsm/kg of water [Figure 1].
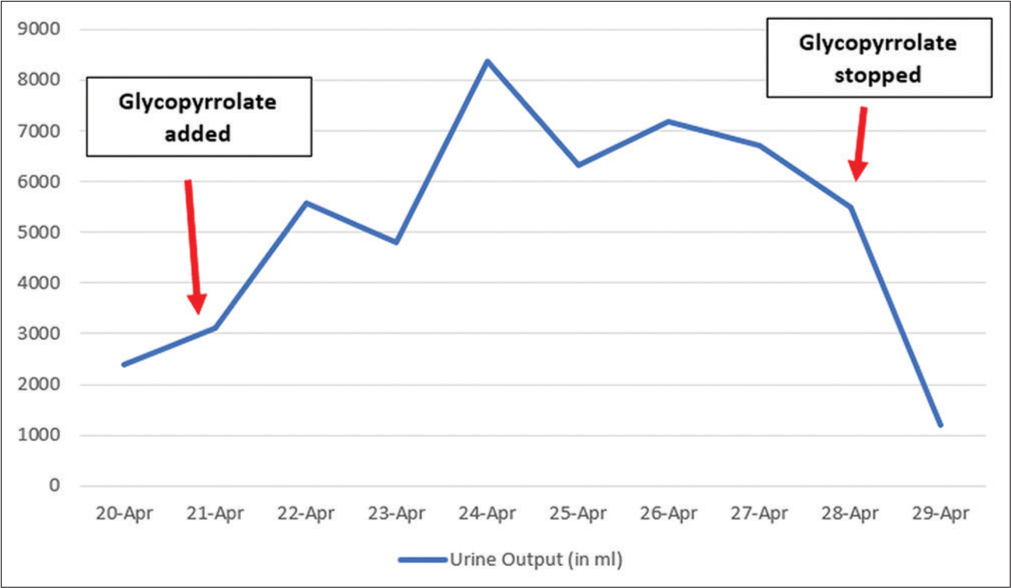
- Excellent temporal relation of glycopyrrolate and polyuria.
DISCUSSION
Broadly, solute and water diuresis are the two main types of polyuria. Most commonly, glycosuria leads to solute diuresis while defective, whereas defective anti-diuretic hormone (ADH) secretion or decreased responsiveness results in water diuresis. In this case, no secondary cause of osmotic diuresis (hyperglycemia, azotemia, or intravenous volume expansion) was found. There was no previous history and current evidence suggestive of neuroendocrine disorder/neoplasms/metastasis, autoimmune disorders, and head trauma. Similarly, there was no history/evidence suggestive of acute renal morbidities i.e., obstruction, acute kidney injury, or the use of diuretics. Potential causes of low urine osmolality, that is, hypokalemia, hypercalcemia, or lithium use, were ruled out.
Previously reported drug-induced polyuria cases occurred secondary to dexmedetomidine, sevoflurane, methotrexate, lithium, cimetidine, and gemfibrozil.[3,4] No reports have been published regarding glycopyrrolate-induced polyuria.
A rare adverse effect of renal impairment documented with IVIG usually develops within 10 days after immunoglobulin infusion and is characterized by oliguria, hematuria, reduced glomerular filtration rate, and elevated serum creatinine. The IVIG has varying concentrations of stabilizing agents, for example, sucrose, maltose, and glucose. Osmotic nephrosis may be the causative mechanism for the renal impairment induced by either maltose or sucrose rather IVIG.[5] In our case, maltose was used as stabilizing agent; still, it is not responsible for symptoms as osmotic nephrosis presents with deterioration in renal functions, and in our case, renal functions were normal.[5]
Syndrome of inappropriate ADH is observed in about 50% of patients with GBS. However, there are few reports of polyuria and transient diabetes insipidus in GBS.[1,6-9] In the case reports, patients were managed by either injection vasopressin/nasal desmopressin; however, in our case, it was resolved spontaneously. The excellent temporal relationship of development and correction of polyuria with the start and gradual stop of glycopyrrolate, respectively, supports our claim that polyuria is attributable to glycopyrrolate.
CONCLUSION
Since this is a commonly used drug in intensive care unit to decrease salivary and tracheobronchial secretions, increased awareness of this uncommon yet significant side effect of glycopyrrolate will encourage physicians to include it during the evaluation of polyuria in inpatients.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent.
Conflicts of interest
There are no conflicts of interest.
Financial support and sponsorship
Nil.
References
- Altered mental status in “GuillainBarré syndrome”-a noteworthy clinical clue. Ann Clin Transl Neurol. 2020;7:2489-507.
- [CrossRef] [PubMed] [Google Scholar]
- Available from: https://www.accessdata.fda.gov/drugsatfda_docs/label/2018/210997s000lbl.pdf [Last accessed on 2022 Aug 10]
- Intraoperative dexmedetomidine-induced polyuria from a loading dose: A case report. J Int Med Res. 2020;48:300060520910643.
- [CrossRef] [PubMed] [Google Scholar]
- Osmotic nephrosis: Acute kidney injury with accumulation of proximal tubular lysosomes due to administration of exogenous solutes. Am J Kidney Dis. 2008;51:491-503.
- [CrossRef] [PubMed] [Google Scholar]
- A case of Guillain-Barré syndrome complicated by diabetes insipidius. Riv Neurobiol. 1968;14:590-605.
- [Google Scholar]
- Transient diabetes insipidus in the Landry-Guillain-Barré syndrome. Arch Neurol. 1972;27:85-6.
- [CrossRef] [PubMed] [Google Scholar]
- Guillain Barre syndrome and polyuria: A rare variant. Jaffna Med J. 2017;29:32-4.
- [CrossRef] [Google Scholar]







