Translate this page into:
Combined central and peripheral demyelination
Address for correspondence: Prof. Bindu Menon, Department of Neurology, Narayana Medical College and Superspeciality Hospital, Chintareddypalem, Nellore - 524 002, Andhra Pradesh, India. E-mail: bn5@rediffmail.com
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
Acquired central and peripheral demyelination in the same patient is a very rare feature. We report a 52-year-male patient with the chronic autoimmune hepatitis (CAH) presenting with pure motor areflexic quadriparesis from 4 months and recent onset of drowsiness of 4 days duration. Studies of imaging and electrophysiology suggested central pontine myelinolysis and chronic inflammatory demyelinating polyradiculoneuropathy. Patient was effectively treated with high dose steroids. To the best of our knowledge, this is the first case of central and peripheral demyelination in a patient with CAH.
Keywords
Central pontine myelinolysis
chronic autoimmune hepatitis
chronic inflammatory demyelinating polyradiculoneuropathy
Introduction
Chronic autoimmune hepatitis (CAH) is a serious hepatic disease with an immunological attack against the liver cells. The disease shows a good response on steroid treatment. Autoimmune diseases result from immune response toward healthy tissue; hence, they can have protean manifestation with multi-system involvement. Central pontine myelinolysis (CPM) is an uncommon condition with demyelination predominantly in the central portion of the basis pontis. Extrapontine areas such as putamen, caudate, thalamus, cerebellum, splenium of the corpus callosum and sub-cortical white matter are also sometimes involved.[12] Osmotic demyelination has been used to describe both central pontine and extrapontine myelinolysis.[3] CPM is generally seen in the setting of rapid correction of hyponatremia. It may also be seen in alcoholics, severe liver disease, after liver transplant, malnutrition, hyperemesis gravidarum and autoimmune deficiency syndrome or toxin exposure. In the past, CPM has been described in cases of chronic active hepatitis.[4] However, no case of central and peripheral demyelination in the setting of chronic active hepatitis has been described and it is essential to recognize the symptoms early to prevent the deficits.
Case Report
A 52-year-old male carpenter, vegetarian by diet, normotensive, non-diabetic and euthyroid presented to neurology department with history of progressive quadriparesis since 4 months. He was diagnosed having chronic active hepatitis 15 months back and was initially started on steroids and was tapered to prednisolone 5 mg/day. He attributed his weakness to hepatic ailment. He attended the neurology department when he developed dysarthria and became drowsy 2 days prior to admission.
On examination, patient was drowsy, but arousable, well oriented to time, place and person and would soon drift back to sleep. On general examination, he had no icterus, flaps and sphincter dysfunction or neck stiffness, but had mild pedal edema. Fundus was normal. Cranial nerve examination showed bilateral eye closure and neck flexor weakness. Tone was flaccid in the limbs; with power proximally 2-3/5 and distally 4/5. His liver function test revealed mild elevation of serum bilirubin 1.7 mg/dl. Full blood count, liver enzymes, coagulation screen, electrolytes, serum ammonia and renal function were normal. He had mild thrombocytopenia with a platelet count of 100,000 lakh/cumm.
Serological testing for hepatitis A, hepatitis B and hepatitis C was negative. Ultrasound abdomen showed shrunken liver with irregular margins and diffusely altered echo texture suggestive of chronic liver disease. Immunological tests demonstrated positive smooth muscle antibody and positive anti-nuclear antibody homogenous staining pattern at a titre of 1 in 6. There was an accompanying hyper-gammaglobulinemia with elevated immunoglobulin G at 2507 (normal range 700-1600) and normal immunoglobulin A levels. Double stranded deoxyribonucleic acid antibodies, anti-mitochondrial antibodies and liver/kidney microsomal antibodies were not detected. Thyroid profile, serum B12, ceruloplasmin, alpha fetoprotein levels, protein electrophoresis was normal; Lupus anticoagulant, P-ANCA, C-ANCA was negative. The positive anti-nuclear and smooth muscle antibody results were strongly suggestive of autoimmune hepatitis and ruled out other autoimmune disorders. Magnetic resonance imaging of brain (T2-weighted) showed hyper intense signals in the basis pontis [Figure 1] suggestive of CPM which accounted for his drowsiness. Nerve conduction studies showed prolonged distal latencies in median (5.5 ms), posterior tibial (6.6 ms) and peroneal nerves (6 ms); decreased motor conduction velocity in median (25 m/s), posterior tibial (30 m/s) and peroneal nerves (41 m/s). F waves were prolonged in median (56), post tibial nerves (70) and peroneal nerves (69). Sensory conductions were normal. The nerve conduction studies were suggestive of chronic inflammatory demyelinating polyneuropathy (CIDP).
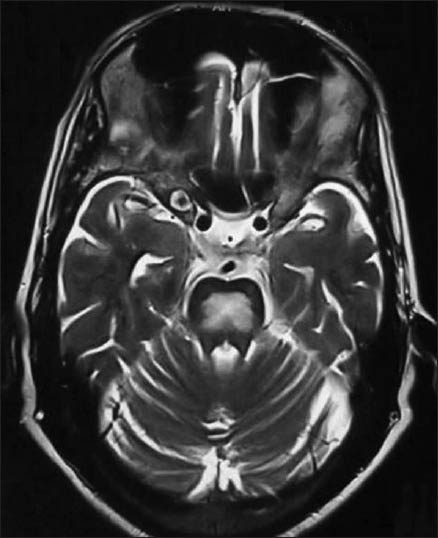
- Hyperintense signal in T2-weighted images in the central pons suggestive of central pontine myelinolysis
With these parameters, patient was diagnosed to have central and peripheral demyelinating disorder. Patient was started on high dose methylprednisolone with meticulous glycemic control. After 10 h of admission, patient sensorium improved and started showing improvement in motor power after 5 days. On discharge, he was able to walk with minimal support on oral corticosteroids. On follow-up after 8 months, he was stable and was maintaining improvement.
Discussion
Our patient was a diagnosed case of CAH. He developed central and peripheral demyelination of equal magnitude. Relationship between the brain and liver has been established since long.[5] The most common neurological involvement is hepatic encephalopathy.[6] Our patient had areflexic quadriparesis, eye closure and neck flexor weakness with sphincter sparing, which was suggestive of CIDP. The same was confirmed electro physiologically. Cerebrospinal fluid analysis was deferred because of thrombocytopenia. The other differential was spinal cord demyelination; however, there was no corticospinal tract involvement clinically as well as on imaging. All the possible secondary causes of CIDP were ruled out by appropriate investigations. As patient was drowsy, impending hepatic encephalopathy was thought. However, he did not have flaps, serum ammonia and electroencephalography was normal. His magnetic resonance confirmed to have CPM. Hence, our patient had both central and peripheral demyelination with equal magnitude.
CIDP is a common form of autoimmune neuropathy with various secondary causes. The underlying cause is possible molecular mimicry between peripheral nerve glycolipids and myelin proteins with various infectious agent components resulting in an immune response to the peripheral nerves. The cause of CIDP is abnormal immune response wherefore the specific triggering mechanism for dysimmune response varies. Our investigation did not reveal any other secondary cause except autoimmune hepatitis. CPM is a form of central demyelination with several causes, but the exact pathogenesis is still not known. Initial description was in alcoholics and malnourished patients.[7] After, it has been described in patients who underwent too rapid correction of hyponatremia. Demyelination has been postulated due to cellular stress, which is the result of fluctuating osmotic forces and ion shifts that lead to changes in cell volume and cell membrane function. Other clinical studies have shown that myelinolysis may be due to the brain's overshoot of sodium during correction of hyponatremia or compression of myelin by edematous cellular elements.[89] The manifestations of CPM may be mild symptoms like behavioral disturbances, drowsiness, confusion, dysarthria, dysphagia, quadriplegia or seizures. Severe cases can have locked-in state or occasionally coma.[210] Our patient had mild symptom, there was no pupillary abnormality as tegmentum was spared. As patient was already quadriplegic, the motor weakness due to the CPM could not be ascertained.
The relationship between the brain and the liver has been known for many years.[5] The most common and most widely recognized is the reversible syndrome of hepatic encephalopathy.[6] CPM and extrapontine myelinolysis are now collected together under the term osmotic demyelination disorder.[11]
These syndromes are not exclusive to liver disease and can occur from a variety of causes,[11] most commonly with alcoholic liver disease. CPM in autoimmune hepatitis has not been reported previously. Our patient was unique as he had both central and peripheral demyelination. Simultaneous central and peripheral demyelination is very rare; though, there have been previous reports in the past.[121314] However, in most of the cases reported there is usually a subclinical involvement of one neuroaxis with more overt involvement of the other. Our patient's clinical picture had involvement of both on admission. Patient responded to steroids. To the best of our knowledge, this is the first reported case of central and peripheral demyelination in autoimmune hepatitis. We wish to report it as treating physicians should be aware of this potential complication. Our patient highlights the fact that in a susceptible individual, both the central and peripheral myelin can be under the immunological attack. Early diagnosis and prompt treatment can prevent further progression in such cases and preventing morbidity and mortality.
Source of Support: Nil.
Conflict of Interest: None declared.
References
- Subcortical white matter lesions in osmotic demyelination syndrome. AJNR Am J Neuroradiol. 1995;16:1495-7.
- [Google Scholar]
- Central pontine myelinolysis: A metabolic disorder of myelin. J Neuropsychiatry Clin Neurosci. 2011;23:369-74.
- [Google Scholar]
- Osmotic demyelination disorders: Central pontine and extrapontine myelinolysis. Curr Opin Neurol. 2000;13:691-7.
- [Google Scholar]
- Central pontine myelinolysis secondary to cytomegalovirus hepatitis in a 10-month-old child. Pediatr Radiol. 2003;33:44-6.
- [Google Scholar]
- A Clinical Treatise on Diseases of the Liver. Translated by Charles Murchison. London: The New Sydenham Society; 1860. p. :193-246.
- [Google Scholar]
- Central pontine myelinolysis: A hitherto undescribed disease occurring in alcoholic and malnourished patients. AMA Arch Neurol Psychiatry. 1959;81:154-72.
- [Google Scholar]
- Decreased diffusion in central pontine myelinolysis. AJNR Am J Neuroradiol. 2001;22:1476-9.
- [Google Scholar]
- Central pontine myelinolysis. In: Rowland LP, Pedley TA, eds. Merritt's Neurology (12th ed). New York: Lippincott Williams and Wilkins; 2010. p. :924-6.
- [Google Scholar]
- Alcohol and the nervous system. In: Neurology and General Medicine (3rd ed). Philadelphia: Churchill Livingstone; 2001. p. :617-29.
- [Google Scholar]
- Acute combined central and peripheral inflammatory demyelination. J Neurol Neurosurg Psychiatry. 2004;75:1784-6.
- [Google Scholar]
- Guillain-Barré syndrome (GBS) with bilateral optic neuritis and central white matter disease. Neurology. 1993;43:842-3.
- [Google Scholar]
- Inflammatory demyelinating polyradiculitis in a patient with acute disseminated encephalomyelitis (ADEM) J Neurol Neurosurg Psychiatry. 1996;60:87-90.
- [Google Scholar]






