Translate this page into:
Cerebrogenic tachyarrhythmia in acute stroke
Address for correspondence: Dr. AS Praveen Kumar, Department of Medicine, Jawaharlal Institute of Postgraduate Medical Education and Research (JIPMER), Pondicherry - 605 006, India. E-mail: jipmer.praveen@gmail.com
This is an open-access article distributed under the terms of the Creative Commons Attribution-Noncommercial-Share Alike 3.0 Unported, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This article was originally published by Medknow Publications & Media Pvt Ltd and was migrated to Scientific Scholar after the change of Publisher.
Abstract
The electrocardiac abnormalities following acute stroke are frequent and seen in both ischemic and hemorrhagic stroke. The changes seen in electrocardiogram (ECG) consist of repolarization abnormalities such as ST elevation, ST depression, negative T waves, and QT prolongation. Among tachyarrhythmias, atrial fibrillation is the most common and occurrence of focal atrial tachycardia is very rare though any cardiac arrhythmias can follow acute stroke. We report a case of focal atrial tachycardia following acute ischemic stroke in 50-year-old female without structural heart disease, and their mechanisms and clinical implications.
Keywords
Atrial tachycardia
atrial fibrillation
insula
ischemic stroke
repolarization abnormality
Introduction
The ECG abnormalities are frequent following acute stroke and occur in as many as 60% to 90% of all stroke patients.[1] The new-onset ECG changes, attributable to acute stroke vary widely in different series and occur in 15% to 30% of cases.[2] ECG abnormalities are more frequent with subarachnoid (SAH) or intracerebral hemorrhage[3] and can also occur in ischemic strokes, following neurosurgical procedures, head injuries, meningitis, epilepsy, and intracranial tumors.[4] The most common ECG changes are QT prolongation and nonspecific ST-T wave changes. The cardiac arrhythmias are also frequent following acute stroke and most common tachyarrhythmia is atrial fibrillation (AF).
Case Report
A 50-year-old female presented with history of sudden onset weakness of right upper and lower limb for 1 day with deviation of angle of mouth to the left side. There was no history of headache, vomiting, seizures, or previous history of stroke or transient ischemic attacks. Also there was no history of chest pain, palpitations, or any cardiac disease. She is not a diabetic or hypertensive and there was no other comorbid illness. Her general examination revealed a pulse rate of 180/ min, regular, low volume, and blood pressure of 130/80 mmHg. The central nervous system examination showed normal higher mental functions, right upper motor neuron facial nerve palsy, and muscle power of 3/5 in upper limbs, 2/5 in lower in lower limbs with right extensor plantar response. The examination of other systems was normal. The laboratory investigations revealed normal liver and renal function tests. The serum calcium, magnesium, and potassium levels were normal. The serum troponin T and creatine phosphokinase levels were normal. The CT brain revealed left middle cerebral artery territory infarct measuring around 2×3 cm.
Our patient presented to the emergency medical services department within 8 h of acute stroke. Her ECG showed regular narrow QRS complex long RP tachycardia at a rate of 186 beats per minute (bpm) with inverted p waves in lead III [Figure 1]. She had a normal study on echocardiography without structural changes or thrombus. On intravenous injection of 6 mg of adenosine, the patient transiently developed complete atrioventricular block but the atrial rate was persisting at the same 186 bpm with regular p waves separated by isoelectric baseline [Figure 2a] which reverted back to AT within a minute [Figure 2b]. A diagnosis of atrial tachycardia was made and she was started on verapamil 40 mg three times daily orally. Following one dose of verapamil she developed sinus bradycardia (50 bpm). So verapamil was withheld and she was monitored in intensive care unit. In the following 1 week, she did not had any other episode of arrhythmia. Considering her elderly age, she was advised to undergo electrophysiological study to know whether she has any substrate for inducible arrhythmia but she did not consent for that. The ECG took before discharge showed normal sinus rhythm (64 bpm), with occasional premature atrial complex. One month later, she came for follow up and she did not report any palpitation and her ECG was in normal sinus rhythm with rate of 68 bpm.
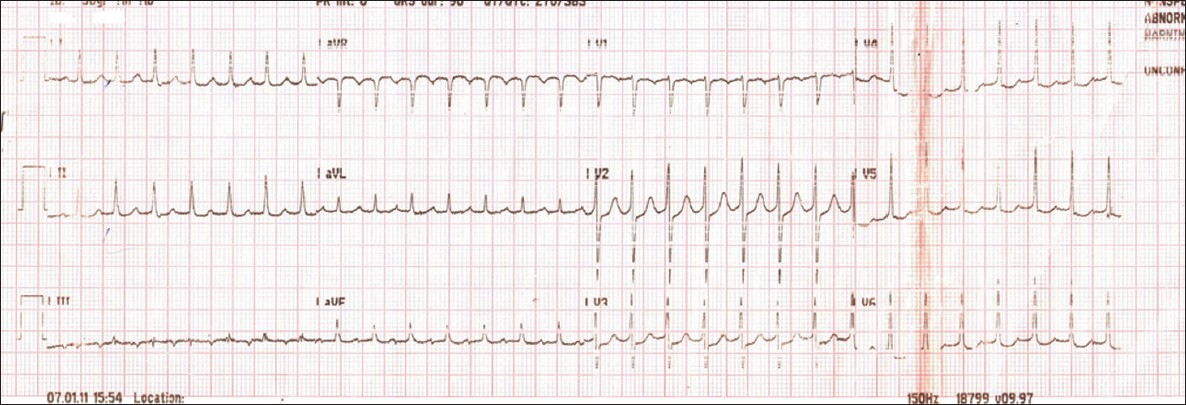
- ECG showing regular narrow QRS complex long RP tachycardia (186 bpm)
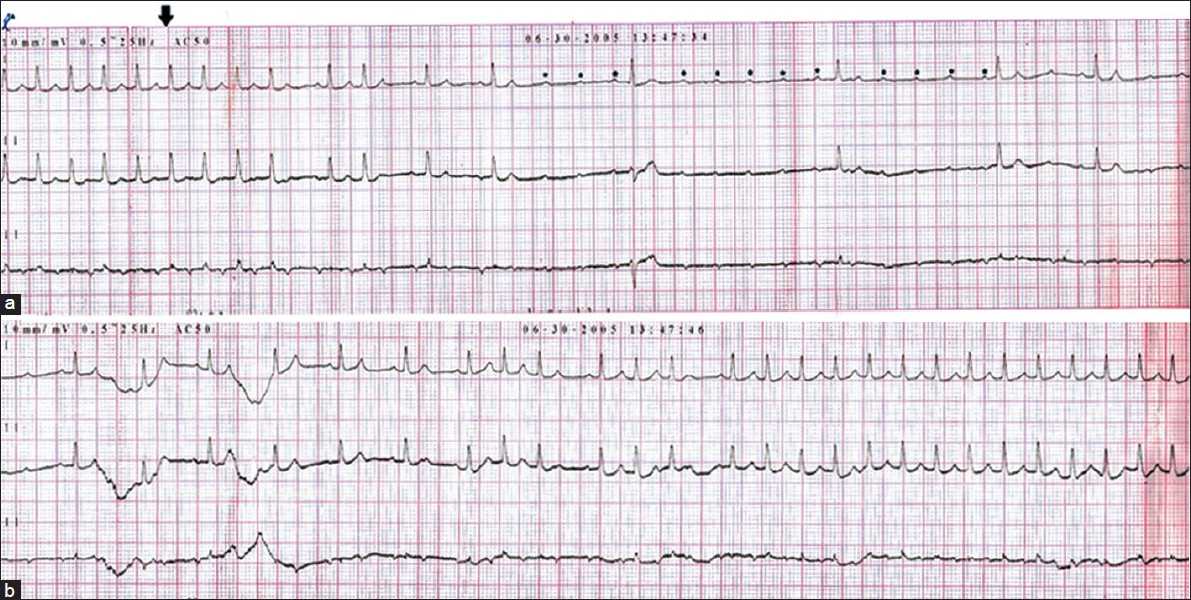
- ECG (a) showing A-V block with atrial rate of 186 bpm with regular p waves (asterisk) following adenosine (arrow) and (b) showing reversion of rhythm to AT after 1 min following adenosine
Discussion
The cardiac arrhythmias following acute stroke include sinus bradycardia, sinus tachycardia, atrial and ventricular extrasystoles, atrial fibrillation (AF)/atrial flutter, and ventricular tachycardia.[4] The exact cause of ECG changes is yet to be delineated and is linked to myocardial injury occurring following acute cerebrovascular pathology.[5] The increased sympathetic tone is attributed to the pathophysiologic mechanism for myocardial damage, via a direct effect or an intense coronary vasoconstrictive influence.[6] This type of cardiac injury is called neural mediated-myocytolysis. The studies[78] have shown increased cardiac sympathetic tone within 5 days of stroke with persistence for at least 3 weeks.[8] The diurnal cardiac autonomic variability was also shown to be disturbed after acute stroke with a loss of nocturnal vagal dominance.[9]
The occurrence of cardiac arrhythmias are more frequent with lesions in the cerebral hemisphere than in the brain stem.[10] The insular cortex and its subcortical connections play an important role in the regulation of autonomic activity and are implicated in the generation of cardiac arrhythmias under stress and following hemispheric stroke. The right insula predominantly controls sympathetic function and the left insula parasympathetic regulation (cardioinhibitory function).[11] It has been shown in humans that left caudal anterior insular stimulation increases the frequency of bradycardia and depressor responses, whereas stimulation of a similar region on right anterior insula is associated with an increase in heart rate and diastolic blood pressure though both types of response were elicitable from either insula.[11]
The AF is the most common tachyarrhythmia and the occurrence of atrial tachycardia is very rare following acute stroke. The study by Norris et al. of 319 stroke patients, 1 patient had paroxysmal atrial tachycardia[10] and Rush et al. reported a case of right frontal tumor with paroxysmal AT[12] which subsided following tumor surgery and no other studies/reports in the literature have documented atrial tachycardia following acute stroke. The mechanisms of AT are reentry, enhanced automaticity or triggered automaticity and all of these mechanisms are activated by increased sympathetic tone. We assume in our case the AT could be due to one of these mechanisms resulting from increased sympathetic tone that follow acute stroke.
The new onset cardiac arrhythmias that follow acute stroke have many important clinical implications. The tachyarrhythmias can result in haemodynamic instability causing reduced cerebral perfusion, which can aggravate stroke and may also increase the risk of cardiac ischemia in patients with associated cardiovascular disease. The arrhythmias such as AF can increase the risk of systemic thromboembolism. The prolonged QTc interval following acute stroke may predispose to polymorphic ventricular tachycardia and sudden cardiac death especially after SAH. The ST-T wave changes that follow stroke can confound with interpretation of ECG changes of myocardial ischemia. Studies have also shown that the long-term risk of cardiac death is increased following stroke.
In conclusion, ECG changes are frequent following stroke and have many clinical implications. The continuous cardiac monitoring of all acute strokes for development of tachyarrhythmia and their timely management can be considered during their initial period of hospital management. Also, as the most common cause of death following acute stroke is cardiovascular death, patients should be on regular periodic follow up.
Source of Support: Nil
Conflict of Interest: None declared.
References
- Electrocardiographic changes in patients with acute stroke: A systematic review. Cerebrovasc Dis. 2002;14:67-76.
- [Google Scholar]
- The cardiac consequences of stroke. In: Barnett HJ, Hachinski V, eds. Cerebral ischemia. Philadelphia: WB Saunders; 1992. p. :167-76.
- [Google Scholar]
- Electrocardiographic patterns in patients with cerebrovascular accidents. Circulation. 1962;25:22-8.
- [Google Scholar]
- Serum cardiac troponin T levels as an indicator of myocardial injury in ischemic and hemorrhagic stroke patients. Tohoku J Exp Med. 2005;205:93-101.
- [Google Scholar]
- Increased intracranial pressure elicits hypertension, increased sympathetic activity, electrocardiographic abnormalities and myocardial damage in rats. J Am Coll Cardiol. 1998;12:727-36.
- [Google Scholar]
- Autonomic nervous activity during sleep in middle cerebral artery infarction. Cerebrovasc Dis. 1998;8:118-23.
- [Google Scholar]
- Location- dependent patterns in cardioautonomic dysfunction in ischaemic stroke. Eur Neurol. 2003;50:30-8.
- [Google Scholar]
- Circadian rhythm of heart rate variability is reversibly abolished in ischemic stroke. Stroke. 1997;28:2150-4.
- [Google Scholar]
- Cerebrogenic cardiac arrhythmias: Cortical lateralization and clinical significance. Clin Auton Res. 2006;16:6-11.
- [Google Scholar]






